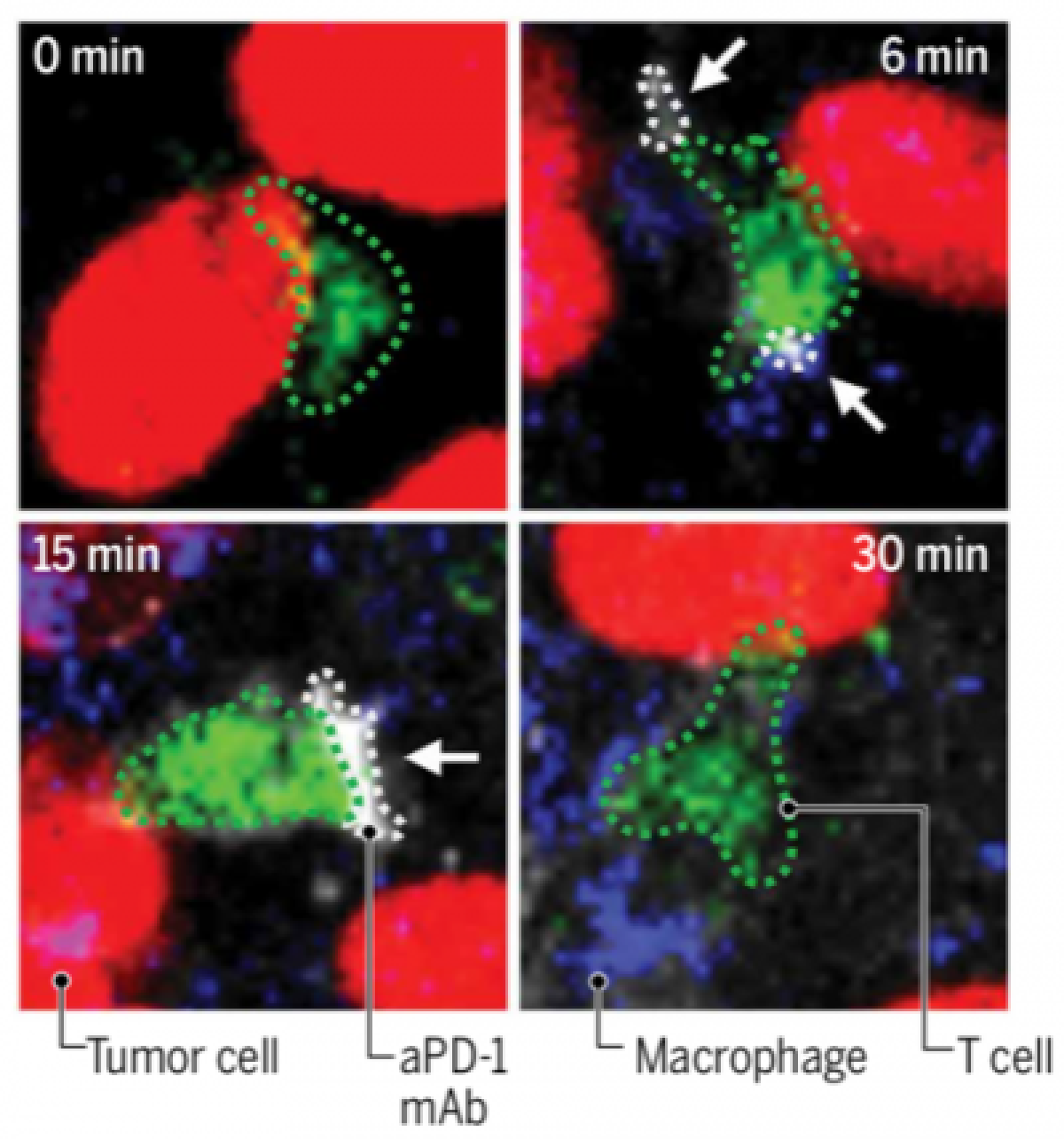
Our groups
The discipline of Biomedical Sciences addresses global challenges such as obesity, cancer, and cardiovascular, autoimmune and infectious diseases. Our programme capitalises on a rich diversity of thematic in genetics, development, microbiology immunology and physiology to offer a stimulating environment to train young scientists toward autonomous and responsible scientific work. The acquisition of fundamental knowledge and a wide range of advanced and transferable skills is achieved through a versatile training program that expands scientific culture and critical thinking. Research projects are embedded in an arena of cutting edge cores facilities that provide personalized training and the proximity of the hospital offers a unique interface between basic and clinical research.

Bacterial glyco-genetics and chemical-genetics
We use cutting-edge glycogenetic and chemical-genetic approaches to unravel two fundamental questions in bacteriology, addressed in (Gram-negative) bacteria. Why and how do bacteria decorate their surface structures such as the flagellum with sialic acids, compared to animals? How is sialic acid covalently conjugated to specific proteins or polysaccharide structures? How are the sialic acids derivatized and how is their synthesis regulated? Another research project deals with the unusual composition of the outer membrane (OM), preventing antibiotic uptake or promoting drug efflux. How is efflux regulated and how is the OM chemically modified? How can multi-drug resistance (MDR) of bacteria can be overcome? Can we modify antibiotics to overcome bacterial MDR?
Apply now

Bone Metabolism
Our laboratory performs translational research on the cellular and molecular mechanisms, and signaling pathways, regulating bone metabolism, and their implication in bone fragility. We are particularly interested in the function and regulation of the WNT/beta-catenin and PDGFR signaling pathways on bone formation and resorption, and in the mechanisms of action and effects of sclerostin inhibitors thereon. Our latest SNF-supported project focuses on bone metabolism and fragility in mouse models of type 1 and type 2 diabetes, to investigate the alterations of osteocytes functions in response to mechanical loading and the role of autophapy, insulin sensitivity, energy metabolism, and MAMS (mitochondria-associated endoplasmic reticulum membranes) in these cells. Another aspect of this grant is a proteomic analysis of sera from subjects with diabetes to unravel new potential mechanisms and biomarkers of diabetic bone disease.
Apply now
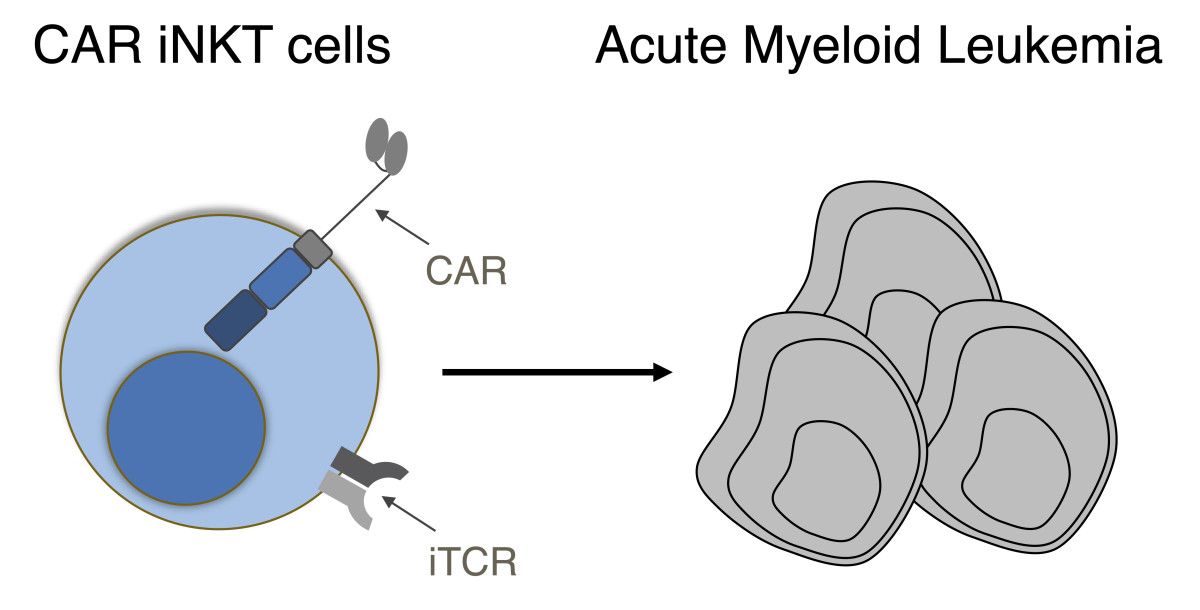
Allogeneic Universal Chimeric Antigen Receptor (CAR) Invariant Natural Killer T (iNKT) Cells Against Acute Myeloid Leukemia
The use of Chimeric antigen receptor (CAR) T cells in acute myeloid leukemia (AML) faces major obstacles. Targeting antigens shared by AML cells and normal myeloid cells leads to bone marrow aplasia. Moreover, autologous T cells are often impaired after intensive chemotherapy. The overall objective of this project is to generate human invariant Natural Killer-T (iNKT) cells against one or several antigens expressed at AML surface. Our central hypothesis is that CAR iNKT cells will exert potent antitumor effects against AML cells through the combinatorial engagement of the CAR and the invariant TCR without inducing myelotoxicity and GvHD. Successful completion of this project will provide pre-clinical evidence for the use of CAR iNKT cells as a potential treatment of AML.
Apply now
Pathophysiology of atherosclerosis
Atherosclerosis and its clinical complications, such as arterial thrombosis, ischemia and myocardial infarction, continue to account for the majority of the morbidity and mortality of the adult population. Atherosclerosis is a progressive, multifactorial process that begins with the accumulation of lymphocyte and/or macrophage-type inflammatory cells in the intimal part of the vascular wall, followed by the proliferation and migration of smooth muscle cells. Our research group is interested in pro- and anti-inflammatory processes and molecules present during the atherogenesis. In different models we study also T and B cell subsets during the early and late stages of atherosclerosis. On another hand, we study the homeostasis of cholesterol through the regulation of LDLR by the endocytosis.
Apply now

Pseudomonas aeruginosa virulence factors and their role in infections
The group is focused on fundamental research into modulating the virulence of P. aeruginosa, the prevention of P. aeruginosa colonization in intubated patients, and the microbiological aspects of colonization or invasion by P. aeruginosa.
Apply now

Surgical reconstruction and tissue regeneration
Our group aims to develop novel and innovative therapeutic strategies for enhancing the clinical outcomes after microsurgical repair and reconstruction of tissues and organs, i.e., neuro-muscular, breast and facial tissues, upper and lower limbs. To transform the surgical reconstructive procedures into multidisciplinary surgical tissue engineering, we combine microsurgical strategies with bioengineering approaches.
Apply now
Diabetes and Cancer
We aim at identifying new molecular targets for better treatment of diabetes and/or cancer.
Apply now

From pathogenesis to precision medicine in women's cancer
In the last decade, large-scale next-generation sequencing of DNA and RNA allowed for a new comprehension of cancer, “the emperor of all maladies” at unprecedented scale. Each tumor is characterized by specific genomic alterations that include multiple gene mutations, amplifications and/or deletions. These cancer cells interact with different components of the tumor microenvironment. Our lab focus on the correlation between genomic alterations of cancer cells, the immune infiltrate and response to therapies in women’s cancer. We are committed to precision medicine by combining our knowledge of genomic alterations of cancer cells and the immune composition of the tumor microenvironment in order to develop new target therapies. We use laser capture microdissection, next-generation sequencing.
Apply now
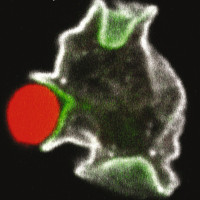
Host-pathogen relationships: recognition, phagocytosis and killing of bacteria.
Phagocytosis is the process by which eukaryotic cells ingest big particles like bacteria. To understand this process, we are making use of a model phagocytic cell, the amoeba Dictyostelium discoideum. Amoebae phagocytose bacteria to feed upon them, and the molecular mechanisms involved are very similar to those observed in mammalian phagocytic cells. Using this simple system, we are studying the role of various gene products in phagocytic cells. Our main aim is to understand how a cell can kill different types of bacteria. Our results indicate that different and largely uncharacterized mechanisms ensure killing of different types of bacteria. Understanding how non-pathogenic bacteria are killed is essential to then study how pathogenic bacteria escape intracellular killing.
Apply now
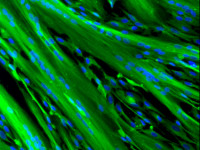
Skeletal muscle regeneration
Our group is studying the mechanisms involved in human skeletal muscle regeneration, using an in vitro system. Skeletal muscle cells are large cells, plurinucleated and unable to divide. Following lesions, the tissue regenerate thanks to satellite cells, the adult muscle stem cells, that are normally quiescent but activate and proliferate as myoblasts after a lesion. This phase is initiated by the inflammation occurring upon injury. Once the inflammation decreases, the myoblasts stop proliferating and start to differentiate, fuse together and repair the damage. The mechanisms taking place during this process include a change of the membrane potential, the activation of calcium signals and transcription factors. Our aim is to understand the interplay between these mechanisms.
Apply now
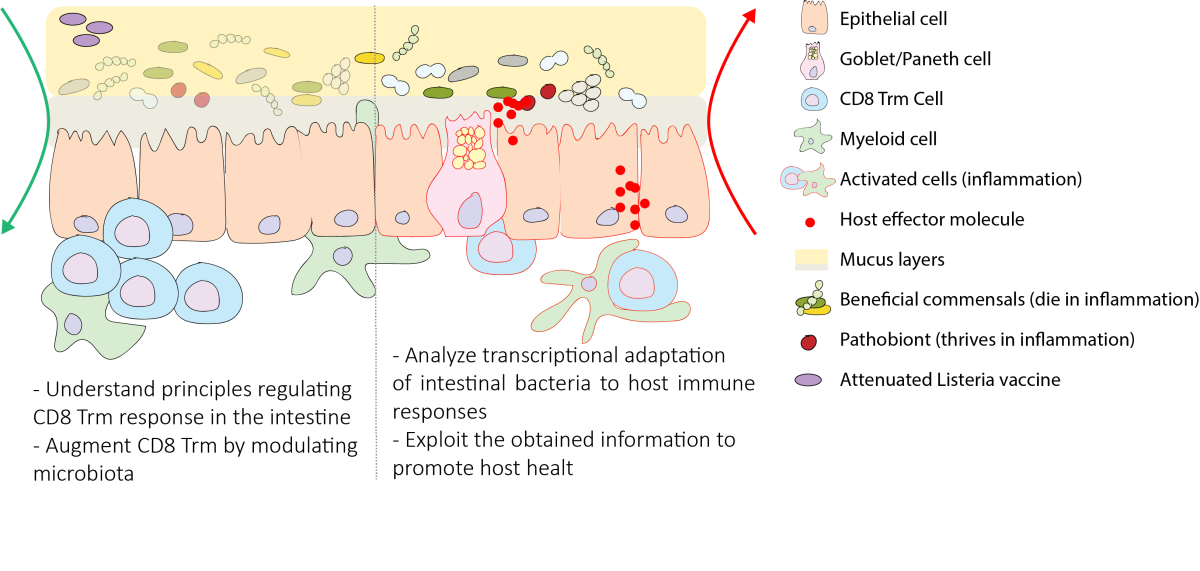
Harnessing the interactions between gut microbiota and immune system
The gut microbiota provides the host with crucial beneficial functions but upon dysregulation can also promote disease. In particular, the gut microbiota is intimately linked to the host immune system, and these two entities can regulate each other’s’ activities through mostly unknown mechanisms. We use a combination of immunological, microbiological and bioinformatics tools to dissect host-microbe interactions at mucosal surfaces and harness them against intestinal disease or favor regional immunity. In particular, our broad goals are: 1) understand how immune responses impact the molecular landscape and functions of beneficial and pathogenic intestinal microbes, and what are the relative implications for host health; 2) potentiate mucosal T cell responses by modulating the gut microbiota.
Apply now
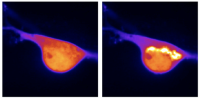
Spatiotemporal Control of Opioid Receptor Signaling
Our lab studies the subcellular organization of opioid receptor signaling in response to chemically distinct opioid ligands. Previously we have shown that opioid drugs differ from endogenous peptides in the cellular location at which they drive receptor activation. It suggests that some drug-specific effects and abuse responses may be explained by what pools of receptors a certain drug is interacting with. Now we aim to unravel how location-specific signaling contributes to opioid drug action and pathology. In this newly established SNSF Eccellenza-funded group, we will combine novel nanobody-based biosensors, live-cell microscopy, neurobiology, pharmacology, and transcriptomic approaches to answer how clinically important signaling systems function on the cellular level.
Apply now

Research in primary care
Our group has projects in two key sectors of primary care research: Clinical and health services research, and research in medical education. We prioritise themes in relation to daily ambulatory practice for which research conducted in hospital environments cannot provide responses. Our main research themes are (1) clinical and health services research: • High quality primary care services for young people • Pragmatic interventions for frequent disorders in primary care • Multimorbidity (2) Research in medical education • Increasing the proportion of medical students choosing a primary care career • Improving clinical teachers skills in primary care • Clinical reasoning (including clinical reasoning in the context of multimorbidity)
Apply now
Molecular and developmental biology of sexual development
The main topic of our research focuses on the study of molecular mechanisms involved in sex determination, differentiation of gonads and testicular function. In particular, we are developing two axes of research: 1) Identification of novel factors involved in disorders of sexual development, and 2) Role of growth factors of the IGF family in mediating testis development and function
Apply now

The inner ear and olfaction laboratory
The inner ear and olfaction laboratory at the University of Geneva is an interdisciplinary research group headed by the clinician-scientist Pr Pascal Senn working on new therapy forms for hearing loss and loss of smell. With respect to hearing, the lab focuses on the two main topics of otoprotection and inner ear regeneration with state-of-the art molecular methods and adequate, preclinical in vivo models. With respect to the sense of smell, the lab focuses on a new approach to induce olfactory regeneration after traumatic loss of the smell function, for which no cure is available today.
Apply now
Glucose metabolism in the diabetic myocardium
Stimulation of glucose transport is one of the first line of defense for the myocardium undergoing ischemic stress. Animal models and clinical studies show that the diabetic myocardium undergoes increased myocardial injury and poorer recovery of function, linked to a reduced capacity to stimulate glucose metabolism during ischemic stress. We investigate cellular mechanisms that in response to the diabetic dyslipidemia may dysregulate metabolic stress-stimulated glucose transport. Complementary in vitro and in vivo experimental approaches are used. Isolated adult rat cardiac myocytes are chronically exposed in vitro to lipids as a model of the diabetic dyslipidemia. In vivo experiments in an animal model of type II diabetes attempt to complement and confirm positive in vitro findings.
Apply now
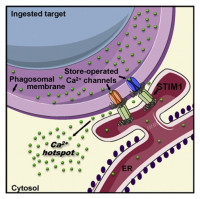
Cell Signaling
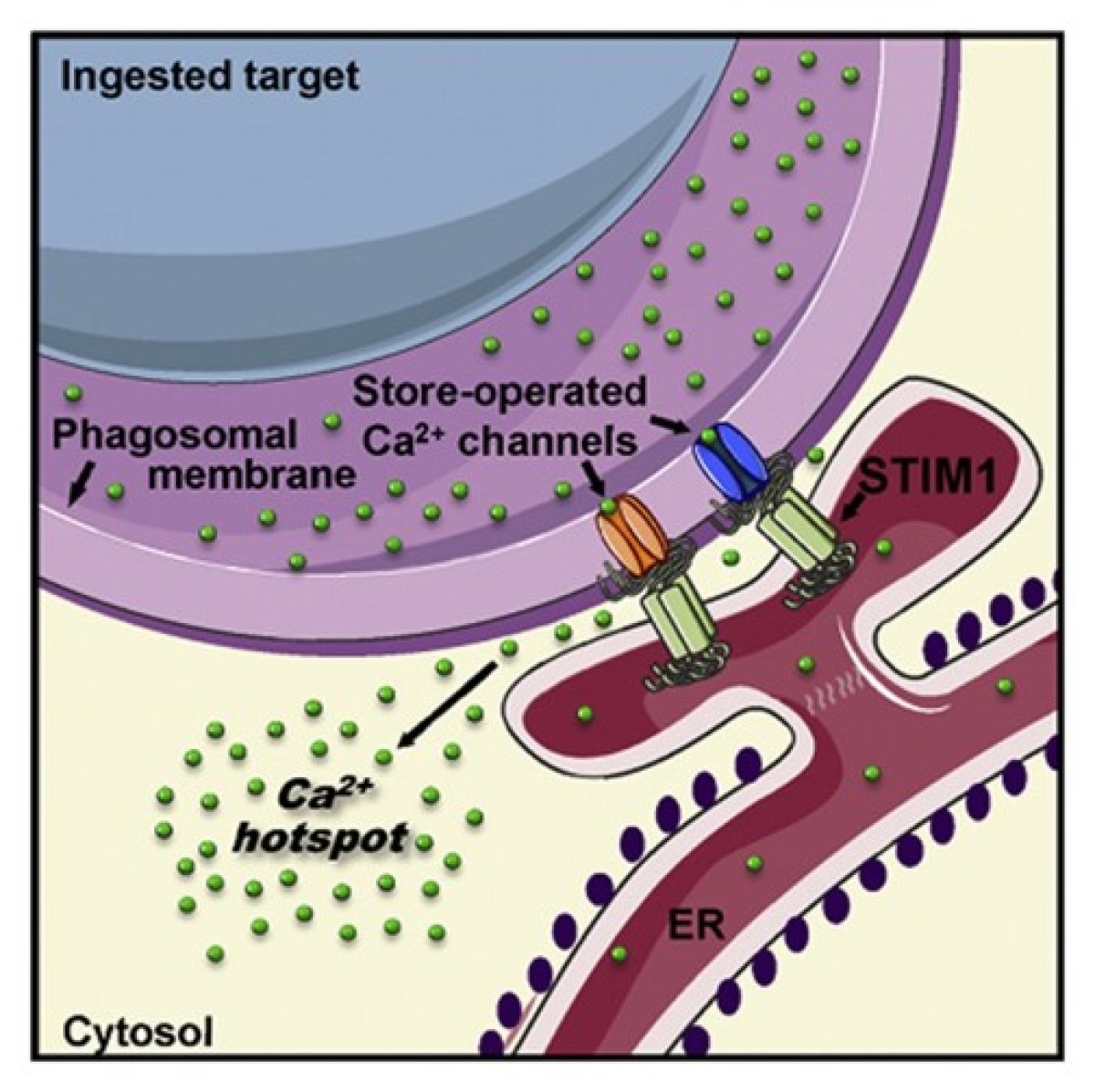
We study the mechanisms controlling the calcium and pH homeostasis of intracellular compartments, with a focus on innate immune cells. We showed that the store-operated calcium entry process mediated by STIM and ORAI proteins sustains the migration and bactericidal activity of phagocytic white blood cells. We now aim to decipher the molecular and ultrastructural determinants of the physiological calcium signals generated at sites of contact between the endoplasmic reticulum and phagocytic vacuoles. For this, we use mouse genetics, ion imaging, electrophysiology, electron microscopy, and a variety of imaging techniques.
Apply now
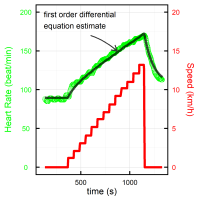
Homeostasis and allostasis of affect: applications to emotional burden of medical decision making
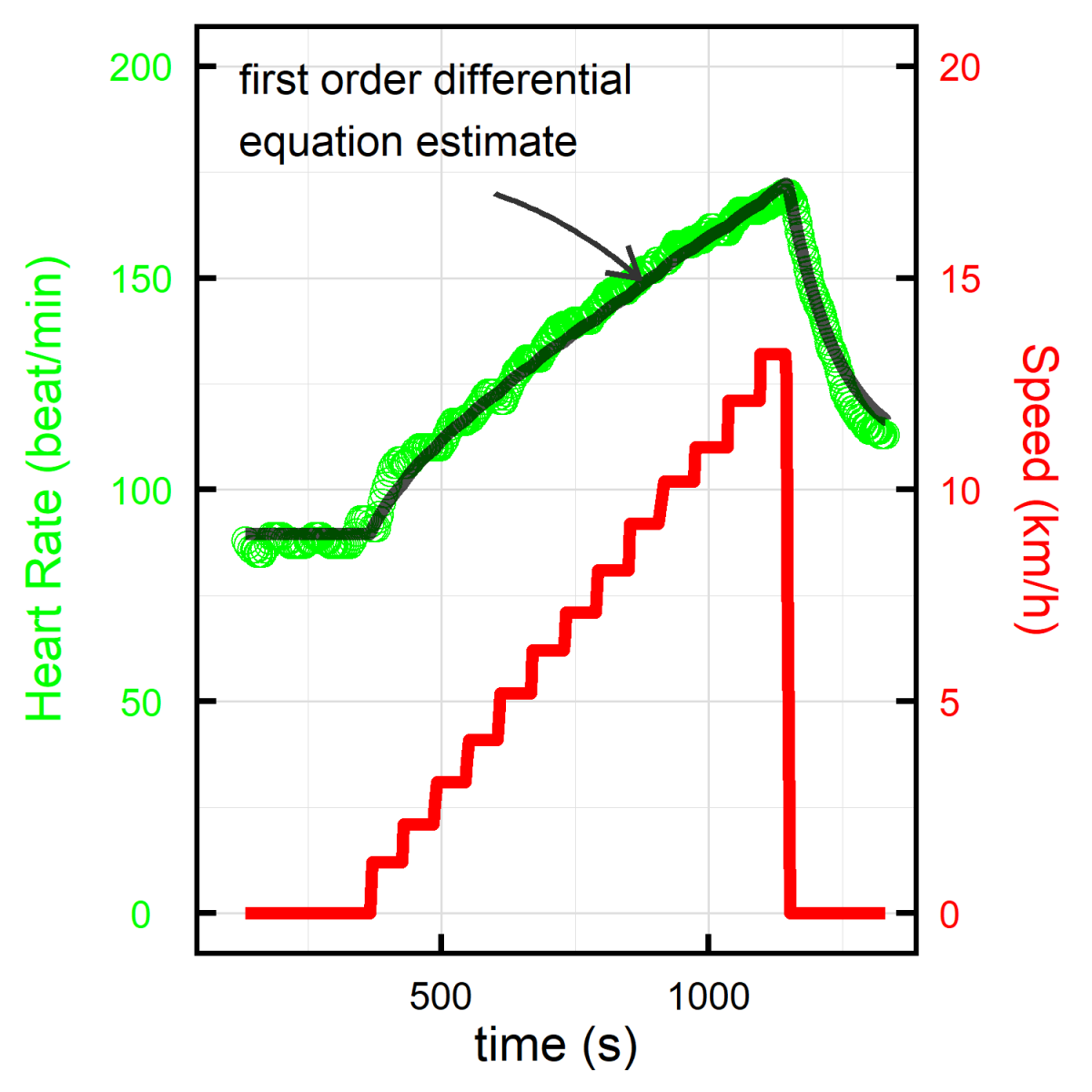
Most healthy processes in biology include a return to equilibrium (homeostasis), potentially with a change in the level of the resting state (allostasis). Our group develop dynamical statistical models to estimate this return to equilibrium for both physiological measures, such as heart rate while doing an effort test, and emotional measures. Applications to heart rate include more precise assessment of training and detraining effects, that can be combined with wearable devices. Applications to emotional measures include evaluating the time and success of dealing with the emotional burden of medical decision making, focusing on the impact on sleep, and intention to change profession.
Apply now
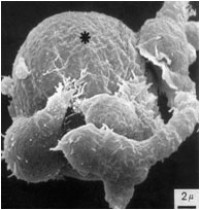
Innate immune cells' responses to antibodies
Immunoglobulin G (IgG) therapies are entering various clinical disciplines, most importantly the fields of oncology, transplantation and autoimmunity. Our current research focus in the general understanding of the mechanisms IgG-therapies; in particular, their effect on Natural killer (NK) cell mediated antibody-dependent cell-mediated cytotoxicity and monocyte (Mo) antibody-dependent cell-mediated phagocytosis.
Apply now
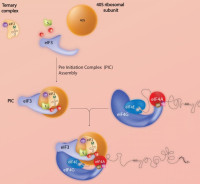
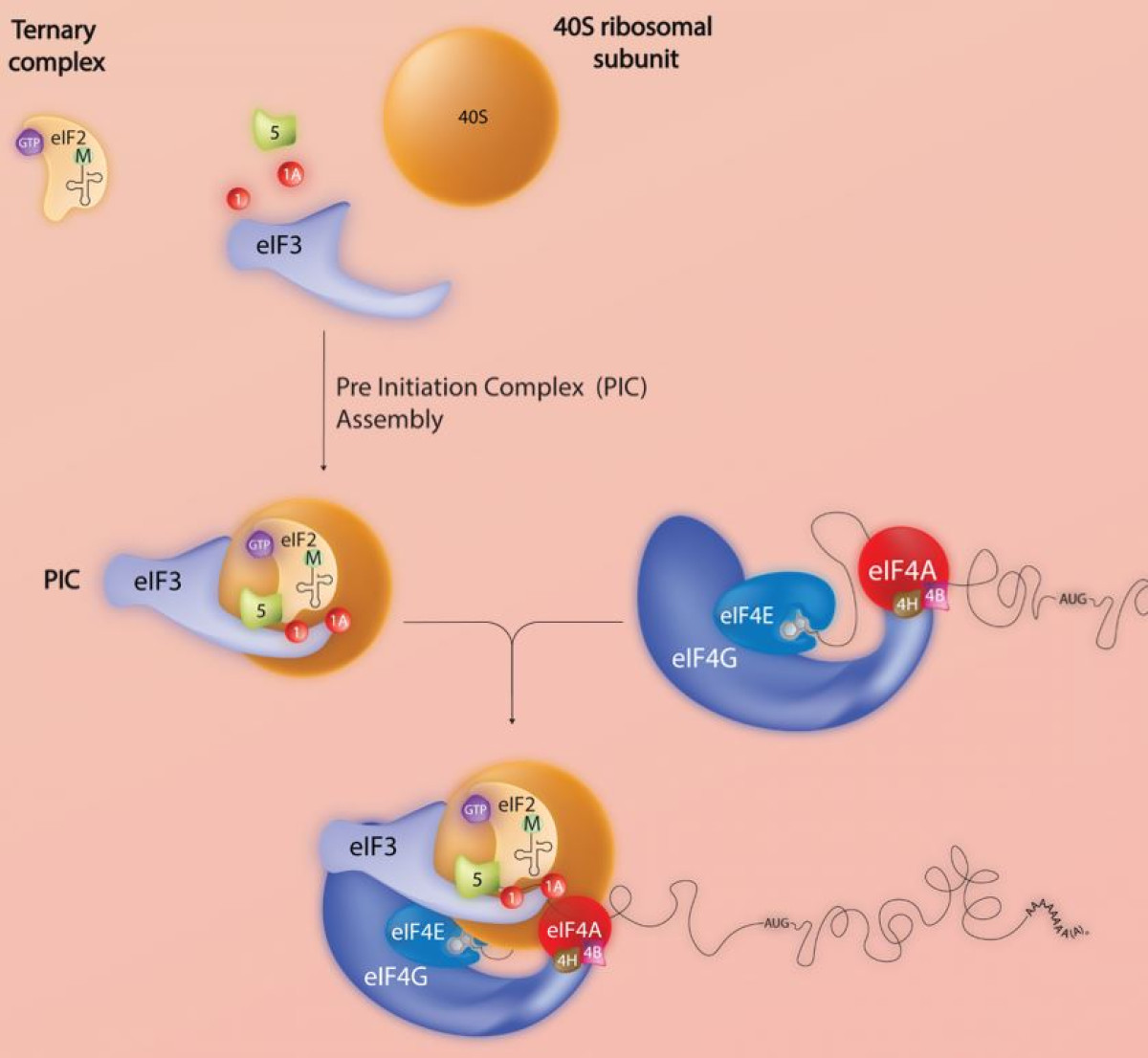
Eukaryotic translation initiation
Protein translation initiation is a key step in the regulation of cell growth. Consequently, dysfunctions of the translational machinery are associated with many human tumours. Our group studies global and gene-specific regulation of initiation in mammalian cells. The objective is to characterise the elements both cis and trans which control the recruitment of mRNAs onto polysomes under defined physiological conditions. More specifically, we are analysing the function of the eukaryotic initiation factor eIF4E3, which binds the 5’ mRNA cap but is not under the control of the 4EBPs. Additionally, using high-throughput polysomal/ribosomal profiling we are exploring the impact of promoter heterogeneity on the protein readout.
Apply now
Translational Brain Biomarkers
Le groupe de recherche s'articule autour de deux axes en neurobiologie a) recherche fondamentale et b) recherche translationnelle intégrant les compétences de chercheurs en biologie, biochimie et chimie, de bio-informaticiens et de cliniciens. L'axe de recherche fondamentale cible la découverte de nouveaux mécanismes dans la neurotoxicité de certains xénobiotiques sur des modèles in vitro de la barrière hématho-encéphalique. L'axe de recherche translationnelle a comme objectif l'identification, le développement et l'implémentation de marqueurs diagnostiques et prognostiques de pathologies cérébrales tels que les accidents vasculaires cérébraux et les traumatismes crâniens.
Apply now
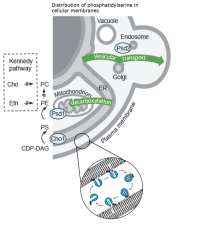
Lipid transport and metabolism
We study how lipid metabolism is organized in time and space, and contributes to the formation of molecular signatures that define organelle membranes or microdomains. Our goals are to understand how this three-dimensional organization is affected during ageing or in some human pathologies, e.g. Alzheimer. We develop original biochemical methods and integrate mass spectrometry, proteomics, lipidomics, microfluidics and bioinformatics to map cellular protein-lipid networks. Current work investigate the role in humans of largely unexplored networks of >100 lipid transfer proteins. How they mediate lipid movements across biological membranes, coordinate lipid metabolism between organelles and signal the (lipid) metabolic needs of each organelle.
Apply now
Immunobiology of brain tumours
My group studies cancer-immune interactions that occur in the specialised microenvironment of the brain. We are exploring how hypoxia impacts on glioblastoma cells, which in turn can modify anti-tumour immunity through modified tumour cell surface phenotype, secretion of immunosuppressive cytokines, or direct communication with immune cells by release of extracellular vesicles. Both T cells and myeloid cells are being investigated as major immune players that can impact tumour progression. We are using new stringent mouse glioma models to test different immunomodulators together with cancer-targeting compounds in order to understand how best to combine different therapeutic modalities for future clinical application.
Apply now

Cellular and molecular analyses of malignant hematologic diseases
The research interests of our group focus currently on three major topics: (A) Study of adhesion molecules (junctional adhesion moelcule (JAM) family) and their role in homing and trafficking of normal hematopoietic cells as well as their role in lymphoma and leukemia development (B) Study of microvesicles, phenotyping and their role in the interaction between tumor cells and the microenvironment (C) Phenotypic analysis of hematopoietic stem cells and minimal residual disease detection in acute leukemia.
Apply now
Pathophysiology of clot structure in thrombosis and bleeding disorders
Fibrin clot structure plays a major role in thrombosis and bleeding. It determines the viscoelastic properties of clot, the resistance to lysis and the interactions with blood cells. Fibrin clot structure is altered in patients with a variety of thrombotic or bleeding disorders. Through a translational approach, combining clinical and fundamental research, our research group investigates the role of fibrin clot structure and function by state-of-the-art techniques. The major aim of our project is to investigate whether and how abnormal fibrin clots contribute to thrombosis and/or bleeding.
Apply now
Sarcopenia, Mobility Disorders, and Falls
Our research group has developed a research line focused on falls, mobility disorders, frailty and sarcopenia in older adults. We are actively continuing our investment in our clearly identifiable research lines on an international level, in collaboration with our extensive network of clinician and researcher experts (local, national, and international) that we have created, as well as conducting numerous clinical studies in parallel.
Apply now
Transplantation and Hepatology
Hepatocellular carcinoma (HCC) is associated with a high mortality, representing the fourth cause of cancer-related death worldwide in 2020. Of all treatment options and for appropriate candidates, liver transplantation offers the best outcome. Our laboratory aims to explore the mechanisms and possible treatments to improve these outcomes. Different fields of investigations are developed in animal models and samples from patients. Our current projects investigate the impact of immune checkpoint inhibitors in liver transplantation for HCC and the role of liver vascularization and portal hypertension on liver diseases.
Apply now
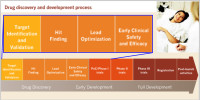
Targeting the Wnt signaling in cancer
The translational direction of our research aims at drug discovery targeting the Wnt signaling pathway in cancer, principally breast cancer. Our pipeline encompasses high-throughput screening, hit-to-lead optimization, and animal PK and PD studies, culminating with early phase clinical trials.
Apply now

Geneva Platelet Group
The Geneva Platelet Group co-directed by Jean-Luc Reny and Pierre Fontana is an interdisciplinary group working with complementary approaches in a truly translational way on the issues of platelet function and antiplatelet drugs. Investigational approaches include basic science, pharmacological, clinical and epidemiological research.
Apply now
Cystic fibrosis and Gap junctions
The lungs of cystic fibrosis patients are chronically infected by opportunistic pathogens. The links between the CF gene (CFTR) defect and the hyper-susceptibility of the CF lungs to stress and infection remains unclear. In this context, we are focusing on gap junctions, the junctional complexes that mediate the direct and regulated transfer of ions, metabolites and second messengers between cells. The regulation of these channels is defective in CF epithelial cells. Our aims are to unravel the intracellular signaling linking CFTR to gap junctions as well as their roles in host innate immunity and abnormal repair in response to infection.
Apply now
The ins and outs of invasion and egress in Apicomplexa
Motion is an intrinsic property of all living organisms and they have evolved diverse strategies to harness cytoskeletal proteins and motor proteins to organise cellular movement. The Apicomplexans are obligate intracellular parasites of considerable medical and veterinary significance that share a unique form of gliding motility, which is essential for their survival and infectivity. We are taking an integrated approach based on reverse genetics, biochemisty and live imaging to elucidate the molecular mechanisms and signaling cascades governing host cell invasion and egress from infected cells by Toxoplasma gondii. In parallel we investigate host-parasite interplays and cyst wall formation during parasite encystation, a process of central importance for pathogenesis and transmission
Apply now
Cell Therapy & Musculoskeletal Disorders
Our research group focuses on the study and improvement of muscle regeneration and the treatment of osteoarthritis. Myogenic stem cell transplantation is a promising approach in the treatment of severe muscle damage as well as in the modulation of inflammation in osteoarthritic patients. We have 2 main areas of research. We want to better characterize, in vitro, human myogenic stem cells in order to improve their survival and their regenerative capacity after transplantation. We also want to study the anti-inflammatory capacities of these cells in experimental models of osteoarthritis in mice.
Apply now
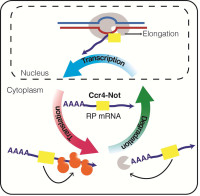
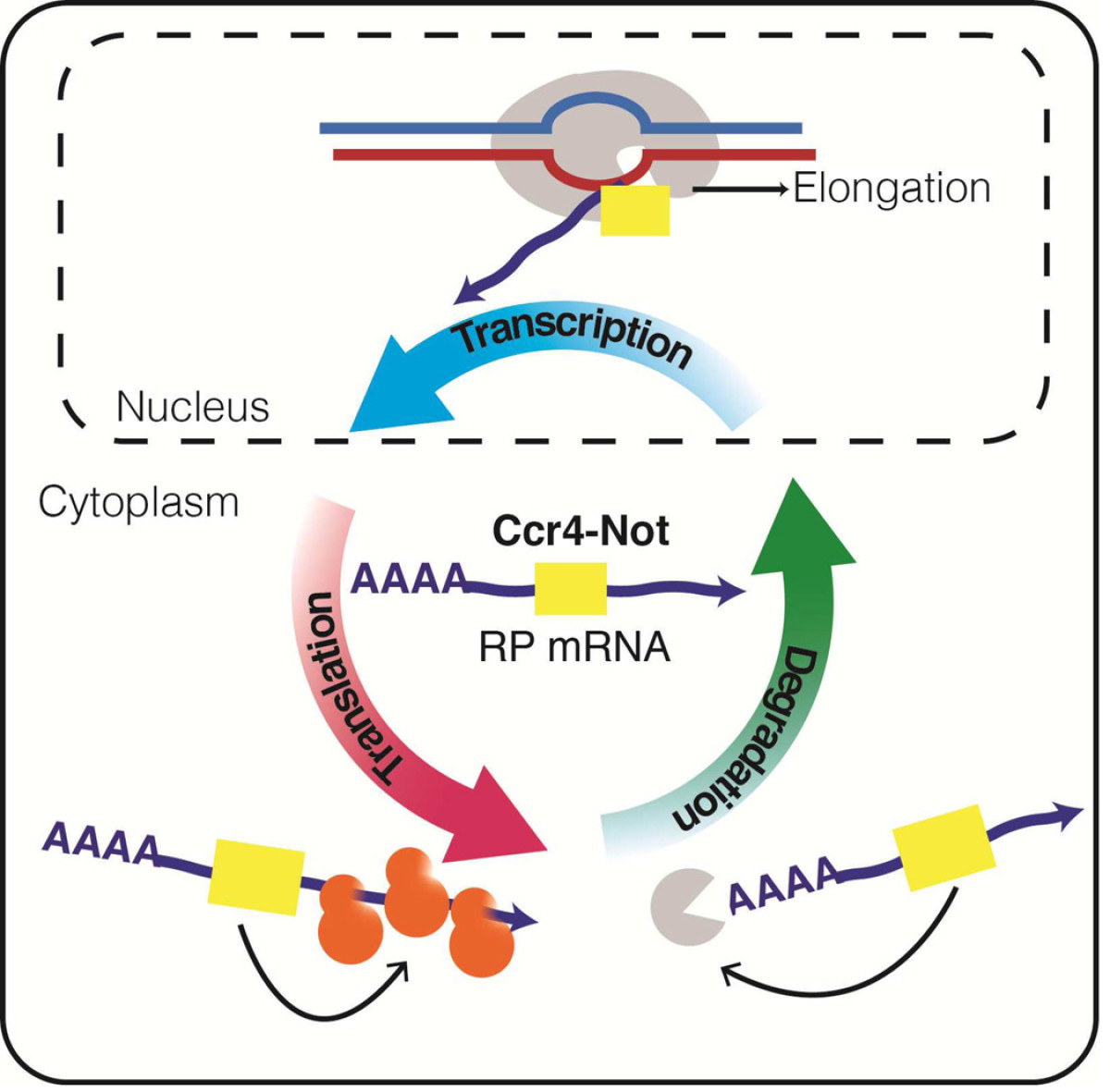
Regulation of Gene expression by the Ccr4-Not complex
The projects of the Collart laboratory deal with the Ccr4-Not complex, conserved across the eukaryotic kingdom and a central regulator of gene expression that acts from mRNA synthesis to degradation of proteins. The Collart laboratory studies this complex regulator in the yeast S.cerevisiae where powerful genetic tools can be combined with biochemical studies. Their working model is that the Ccr4-Not complex works as a "chaperone platform", regulating dynamic functional interactions between cellular partners. By interacting with translating ribosomes and chaperones, it has the ability to interact with newly synthesized proteins and "deliver" these proteins to their cellular partners in a functional way. The projects of the Collart laboratory are targeted towards addressing this model.
Apply now

Individualised Therapy in Pediatric Oncology and Hematology (Pharmacogenomic)
The CANSEARCH research laboratory has more than 10 years’ experience in pharmacogenomics research and is collaborating internationally in this field. The laboratory’s expertise includes the application of pharmacogenomics research to tailor drug dosing in pediatric cancer treatment and to apply genetic models to predict outcomes in various pediatric cancers and in the hematopoietic stem cell transplantation (HSCT) setting. The laboratory currently has different pharmacogenomics studies as principal investigator, including one open in 20 countries (Polymorphism and Busulfan Pharmacokinetic study in Acute lymphoblastic leukaemia (ALL SCT ped Forum study; clinicaltrial.gov ID: NCT01949129; NCT02670564) , as well as in the pharmacogenomics study in myeloid leukaemia.
Apply now
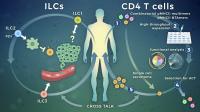
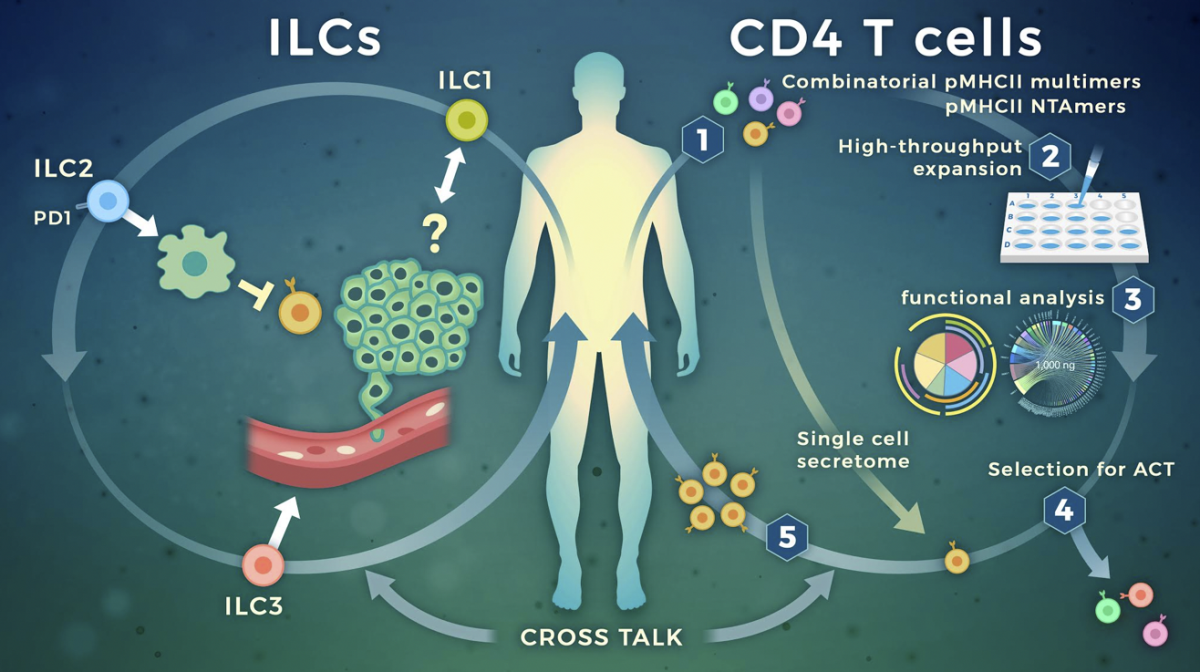
Targeting cytokine secreting lymphocytes for cancer immunotherapy
Our research focuses on dissecting the role of innate lymphoid cells (ILCs) and tumor-specific CD4 T cells in anti-tumor immunity, to optimize current immunotherapy strategies. By combining studies directly in patient’s samples with analyses in relevant tumor mouse models we explore how cytokine secreting lymphocytes interact with tumor and stromal cells to sculpt the tumor microenvironment. Using transcriptomic, epigenetic and phenotypic studies down to the single cell level we are assessing quantitative and qualitative alterations of cytokine secreting lymphocytes directly in cancer patients, pre- and post-treatment. By interfering in vitro and in vivo with identified targetable pathways we are exploring therapeutic options that exploit these cells for therapy.
Apply now
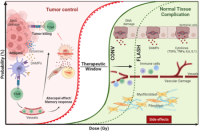
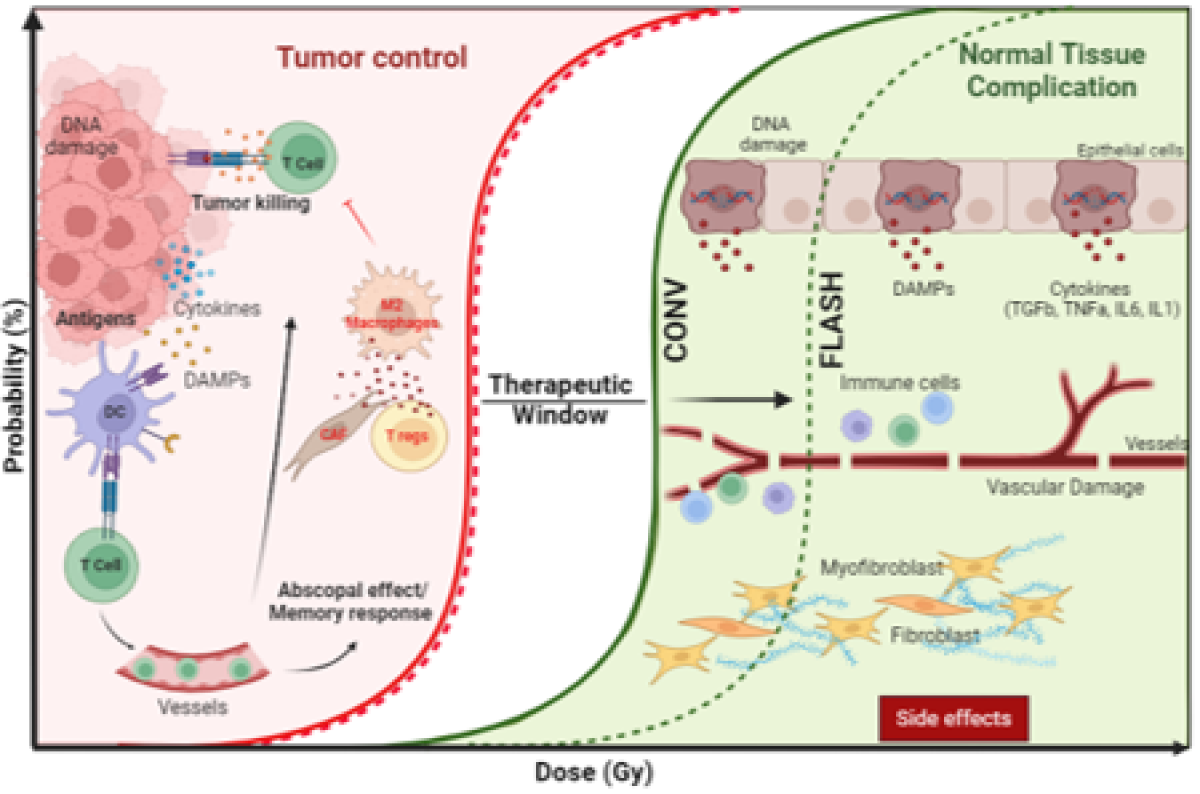
LiRR: Laboratory for innovation in Radiobiology applied to radiotherapy
Radiotherapy (RT) is one of the most effective and cost-effective modalities for treating cancer and today more than half of all cancer patients will receive RT during their treatment. Thanks to technological and radiobiological improvements, RT is today a precise and conformational treatment. However, the treatment of radioresistant tumors remains limited by the tolerance of healthy tissues, which prevents increasing the RT dose and reaching a curative threshold. Thus, enhancing the differential effect of RT between tumor and healthy tissue, either by radiosensitizing the tumor or protecting healthy tissues selectively, remains the overarching goal in radio-oncology. Our laboratory develops translational research in radio-oncology to transfer advanced RT strategies to the clinic. This program aims at offering innovative treatments for our patients and strengthen multidisciplinary interactions amongst the fields of radiobiology, physics, adult and pediatric oncology, imaging, and pharmacology. FLASH Radiotherapy: Our group pioneered FLASH radiotherapy which is a breakthrough in radio-oncology based upon the administration of irradiation in an extremely short time (milliseconds or less) and at ultra-high dose rates (greater than 100 Gy/s), as opposed to standard conventional radiation which is delivered over several minutes at conventional dose rates (dose rates of 0.1 Gy/s). The main advantage of FLASH is to induce similar tumor control and reduce normal tissue complications compared to standard RT at conventional dose rate. While continuing our studies with protons and VHEE in collaboration with PSI and CERN, we are developing at HUG/UNIGE a new hub dedicated to X-rays for conformational FLASH radiotherapy. Our work primarily focuses on brain tumors, particularly rare pediatric tumors requiring special attention, and is funded by 7 academic grants. Personalized Radiotherapy: Focus on Breast Cancer Today, breast cancer is no longer considered a single disease but rather a set of different diseases. The understanding and classification of the disease at the molecular level have brought enormous benefits in overall survival and quality of life for patients, allowing for the personalization of treatment. Our goal is to personalize breast cancer radiotherapy based on its heterogeneity, which is considered today as the main challenge in innovation in breast radiotherapy.
Apply now
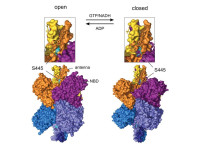
Mitochondria and energy metabolism
We study how mitochondria integrate and generate metabolic signals controlling the coupling of glucose recognition to insulin exocytosis. Current work investigates generation, intracellular transport, and mechanism of action of glutamate in beta-cells and brain; related to energy homeostasis. We are particularly interested in the Glud1 gene that encodes for glutamate dehydrogenase. Beside tissue-specific knockouts, we study Glud1 activating mutations giving rise to hyperinsulinemia (beta-cell), hyperammonemia (liver), and epilepsy (brain).
Apply now
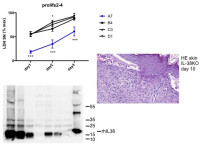
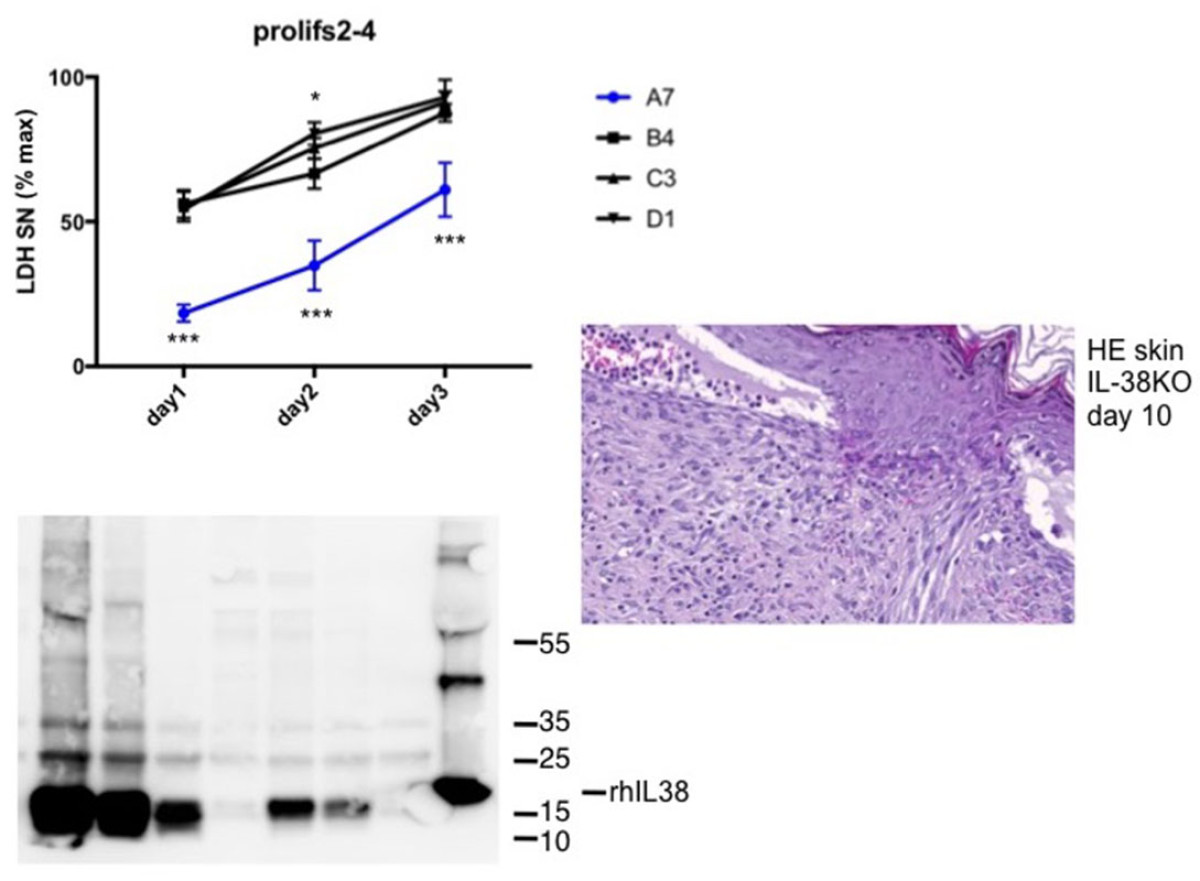
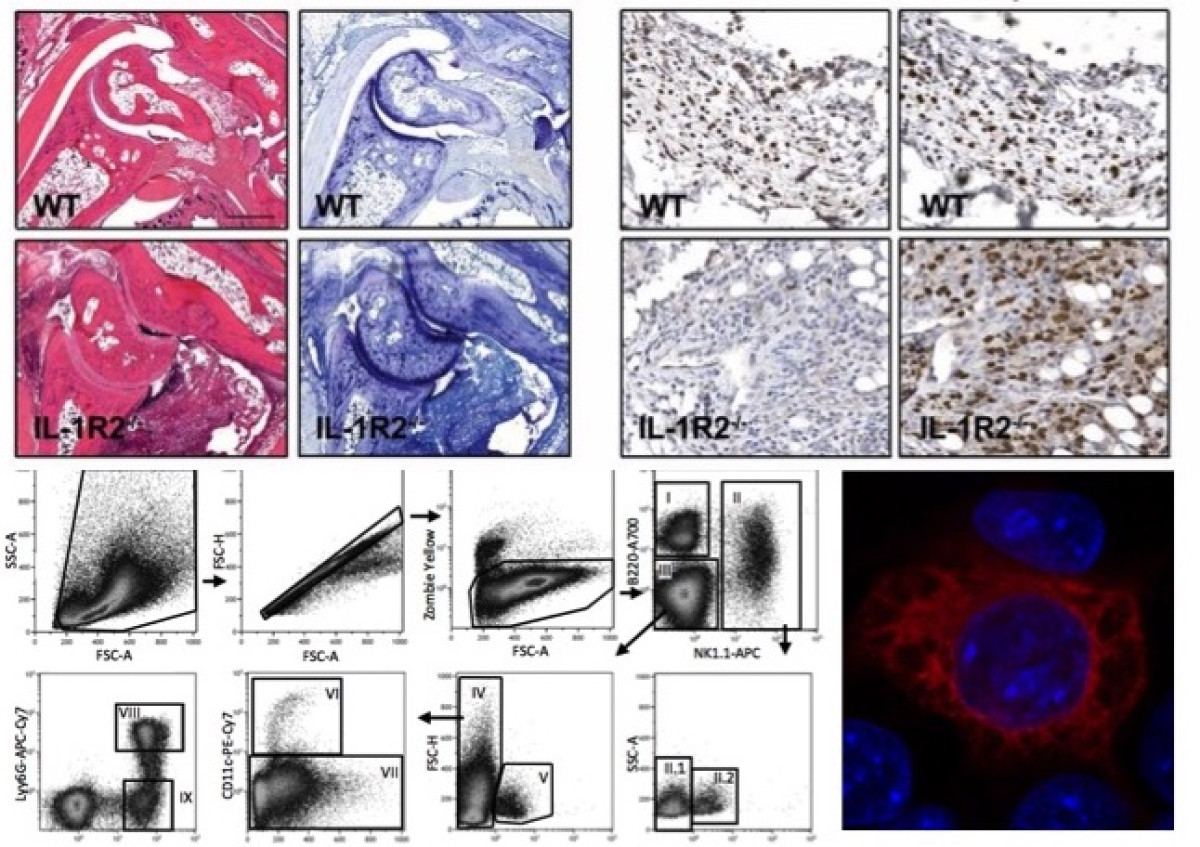
Biology of interleukin-38
The interleukin (IL)-1 family includes seven pro-inflammatory cytokines and four members with established or hypothetical antagonist activity, including IL-38. Agonistic IL-1 family cytokines have specific roles in innate and adaptive immunity, while antagonists limit uncontrolled inflammation and immune responses. Little information is available to date concerning expression and function of IL-38. Our working hypothesis is that IL-38 acts as a naturally occurring inhibitor of inflammation and might possess therapeutic properties in specific immune-mediated inflammatory diseases. The aim of this project is to further study the biology and potential anti-inflammatory properties of IL-38 using cultured human and mouse cells and different mouse models of disease.
Apply now
Cell adhesion and migration in health and disease
Cell adhesion between cells or to the extracellular matrix is essential to maintain the healthy state of a tissue. During development, but also in pathologies such as cancer or fibrosis, alterations in the dynamic remodeling of the cell-cell and cell-extracellular matrix contact leads to the formation of new tissues and organs, but will also lead to life-threatening pathologies. By using a structure-function approach we study the structural assembly of integrin-containing cell-matrix adhesion sites, and we would like to understand how they are regulated by mechanical tension, by receptor tyrosine kinases signaling and by the metabolic status of cells. Fluorescent proteins and live-cell imaging are used to track integrin receptors and their adapters to identify new therapeutic targets.
Apply now
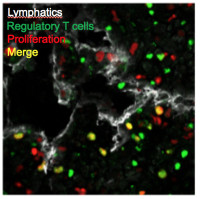
Role of unconventional antigen presenting cells in Autoimmunity and Cancer
Our projects study the contribution of unconventional antigen presenting cells, such as plasmacytoid dendritic cells or lymph nodes stromal cells, to peripheral T cell responses. Depending on the immune microenvironment, those cells can function either as tolerogenic or immunogenic antigen presenting cells. Our research aims at identifying and targeting new cellular functions to promote or in contrast inhibit T cell tolerance in the treatment of autoimmune diseases and cancer development, respectively.
Apply now
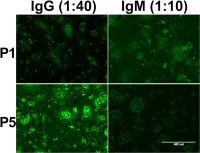
In-depth characterization of antibody responses to vaccination and viral infection
Antibody responses after vaccination or infection can be heterogeneous in individuals and between different viruses or vaccination strategies. In the best case scenario infected or vaccinated patients are protected life-long from disease (e.g. yellow fever or measles vaccination). However, other outcomes such as short-term or sub-optimal protection (influenza vaccination) or even exacerbated infection through antibody-dependent enhancement (dengue virus infection, vaccination with formalin-inactivated RSV) are possible. My group aims to better understand the role of antibodies in infection prevention by characterizing the individual antibody repertoire through antibody quantification, epitope mapping and functional analysis by affinity measurement and neutralization.
Apply now
Role of heterotypic interactions in the function of pancreatic islets
Pancreatic islets are characterized by a high number of heterotypic intercellular contacts between alpha and beta cells. Many studies indicate that cell interrelationships within islets are essential in the function of the endocrine pancreas. However whether and how direct contacts between alpha (secreting glucagon) and beta cells (secreting insulin) are involved in the hormonal secretion of these cells is poorly understood. The alpha and beta cells have a function per se in regulating glucose homeostasis, but through different mediators they secrete and/or via direct intercellular contacts may also regulate the function of the neighbouring cells. The major aim of our study is to investigate whether and how direct and/or indirect heterotypic contacts between alpha and beta cells contribute
Apply now
Signalling in malaria parasites
Malaria is a profound human health problem that kills almost half a million people each year. It is caused by parasites that replicate inside red blood cells and that can only be transmitted by mosquitoes. To time and adapt their development, malaria parasites use a complex system of intracellular signalling networks. We want to understand how signalling networks regulate the development and transmission of malaria parasites so that they can be targeted by new drugs.
Apply now
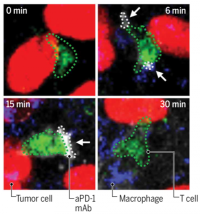
Cancer Immunity and Immunotherapy
We aim to acquire missing knowledge needed to find better ways to fight cancer. To this end, we combine fundamental and translational studies, and focus on harnessing the immune system.
Apply now
RNA metabolism and bacterial pathogenesis
Ribonucleic acid (RNA) is a central molecule for life displaying a great variety of cellular functions in all living organisms. In bacteria, RNA stability varies significantly in response to external conditions, allowing to quickly adapt gene expression during environment or host colonization. In our group, we use the opportunistic pathogen Pseudomonas aeruginosa as model organism to understand how RNA metabolism contribute to bacterial pathogenesis. P. aeruginosa is one of the leading causes of nosocomial infection worldwide and a concern for future healthcare provision due to its antibiotic resistance. By applying genetics, biochemistry, and imaging techniques we aim at characterizing novel RNA-binding proteins and RNA-based regulatory mechanisms affecting P. aeruginosa virulence.
Apply now

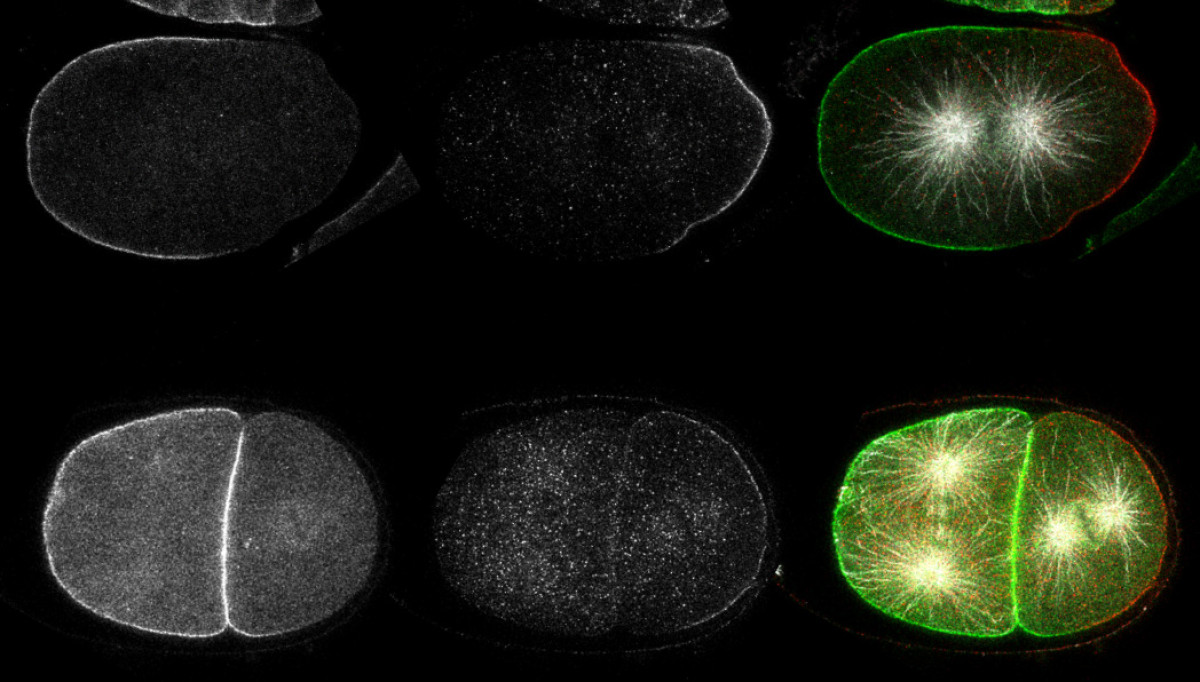
Asymmetric cell division
A human body is made of approximately 10^13 cells. All these cells arise through cell division starting from a unique cell, the fertilized oocyte. Cell division is therefore a crucial process that is required for an organism to grow. A specialized cell division, asymmetric cell division, contributes to cell differentiation, an essential process during development and in adult life. We study the processes of cell division and asymmetric cell division using human cells and the embryo of the roundworm Caenorhabditis elegans as model systems. We focus on understanding how cells are polarized and how the mitotic spindle, the structure the segregates the genetic material between daughter cells, is assembled and properly oriented during asymmetric cell division.
Apply now
Brain inflammation
Our research group focuses on inflammatory processes in the central nervous system (CNS). Using model systems of human inflammatory brain diseases as well as human tissue samples, we investigate immunopathological processes in autoimmune inflammatory processes, as can be observed in multiple sclerosis. Furthermore, we investigate the formation and role of immunological memory cells in the context of viral infections of the CNS.
Apply now
Cellular stress responses to virus infection
Our research focuses on understanding how cells respond to the onslaught of virus infections through intricate cellular stress responses. Viral infections disrupt normal cellular functions, leading to the activation of stress-induced signaling pathways such as the unfolded protein response, that limits viral spread and protect cellular machinery. However, viruses have evolved mechanisms to manipulate these stress responses to their advantage, modifying cellular environments to facilitate replication and evade immune defenses. Using state-of-the-art imaging-based and biochemical approaches, we aim to understand the intricacies of these co-evolved mechanisms to find new cellular functions and minimize virus-induced cellular damage to ultimately contribute to the development of antiviral strategies.
Apply now
Regulation of cell-cell fusion
The syncytiotrophoblast (STB) is the outer layer of the foetal part of the placenta which is in direct contact with maternal blood. It is the main site of exchange for nutrients and gases between the mother and the fetus. Since their nuclei are not able to replicate, cytotrophoblast (CTB) fuse with overlying STB to bring fresh cellular components to the STB. To maintain homeostasis, apoptotic material of STB is packed into syncytial knots and then released into the maternal circulation. An increase in syncytial knots release could induce an inflammatory response by the mother and may lead to preeclampsia (PE), a major cause of maternal mortality and morbidity. Controlled CTB-STB fusion and trophoblast turnover are thus crucial to maintain the integrity of placental barrier.
Apply now
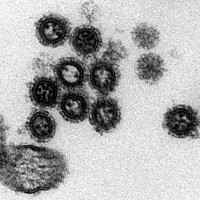
Influenza A virus pathology
Viruses are highly adapted to their host organism in order to replicate successfully. On a molecular level my team works on understanding the interplay of virus and host factors within the host cell and develops tools to identify new host proteins, which are either required for or which counteract virus replication. On organism level we are addressing how acute viral infections, e.g. with influenza A virus, disturb the fine-tuned balance of microbial communities in our body, and how these disturbances affect short and long-term physiological functions of the host.
Apply now
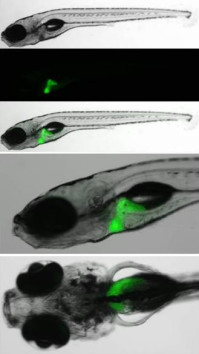
Translational Research on Disorders of Hemostasis and Thrombosis
We are studying genetic disorders of hemostasis and thrombosis, applying the results of whole exome sequencing and functional analyses in transfected cells, zebrafish animal models and patient samples towards a personalised approach to treat these disorders. Our main interest is in fibrinogen deficiencies, both quantitative and qualitative. We investigate the molecular mechanisms by which fibrinogen gene mutations lead to disease and other mechanisms influencing the variable phenotypes of patients, including modifying gene polymorphisms. Finally, we are studying novel mechanisms determining fibrinogen levels in the circulation, a project which is clinically relevant since a high fibrinogen level is an independent risk factor for cardiovascular disease.
Apply now
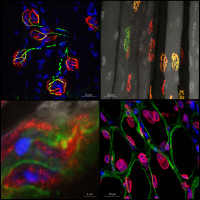
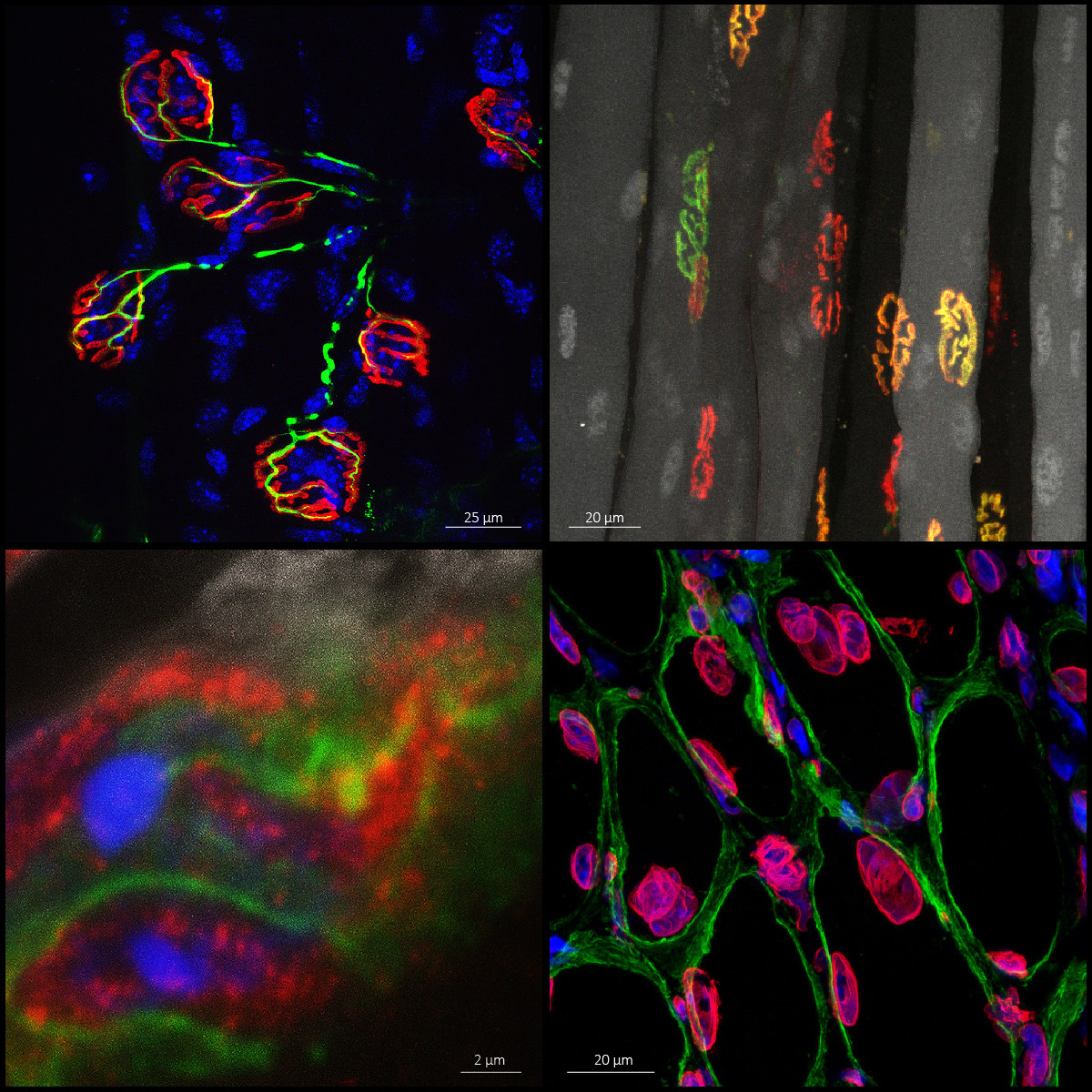
Muscle and NeuroMuscular Junction
Skeletal muscle dysfunction marks the decline of patient’s health condition in numerous pathological conditions. This often correlates with the loss of functional innervation of the tissue. The group focuses on the molecular network involved in the maintenance and plasticity of neuromuscular junctions in muscle. Specifically, we investigate the mechanisms regulating the expression and dynamics of synaptic proteins and the events triggering synaptic remodeling in muscle fibers. In translational projects, we further examine the processes threatening the neuromuscular system in specific disorders. We benefit from state-of-the art procedures to analyze signaling pathways and cellular processes in vivo using mouse models, in vitro using mature muscle cells, as well as in human muscle biopsies.
Apply now

Emerging viral infections in humans and animals
The Geneva Centre for Emerging Viral Diseases, where I am based, is a joint institution of the University Hospitals of Geneva and the University of Geneva - with the aim to understand and prevent the spread of emerging and re-emerging viruses, by providing diagnostic capacity combined with clinical expertise and basic research. My research interests in particular are the laboratory-based risk assessment of zoonotic viruses. This includes the development of epithelial cell culture models from reservoir hosts such as bats and rodents and the assessment of viral diversity and virus discovery in both humans and animals. Further research interests are in the field of clinical virology, such as diagnostics for zoonotic viruses and assessment of imported virus infections.
Apply now

Molecular mechanisms of skin aging
Our laboratory has been mainly interested in the role of CD44 and its interaction with hyaluronate (HA) in skin aging. CD44 is a polymorphic transmembrane glycoprotein and the principal cell surface receptor of HA. We have observed a decrease of epidermal CD44 and HA in dermatoporosis, a term that we proposed for a chronic cutaneous insufficiency/fragility syndrome of the elderly. We have shown that dermatoporosis is due to the dysfunction of hyalurosome which is a putative multimeric macromolecule complex composed of molecules involved in HA metabolism and cell signaling in keratinocytes, such as CD44, HB-EGF and erbB1. According to our studies, epidermal Lrig1+ stem cells, Wnt/-catenin pathway, calcium signaling and p16Ink4a may also play a role in the pathogenesis of dermatoporosis.
Apply now
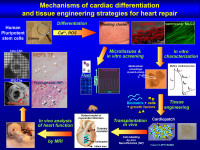
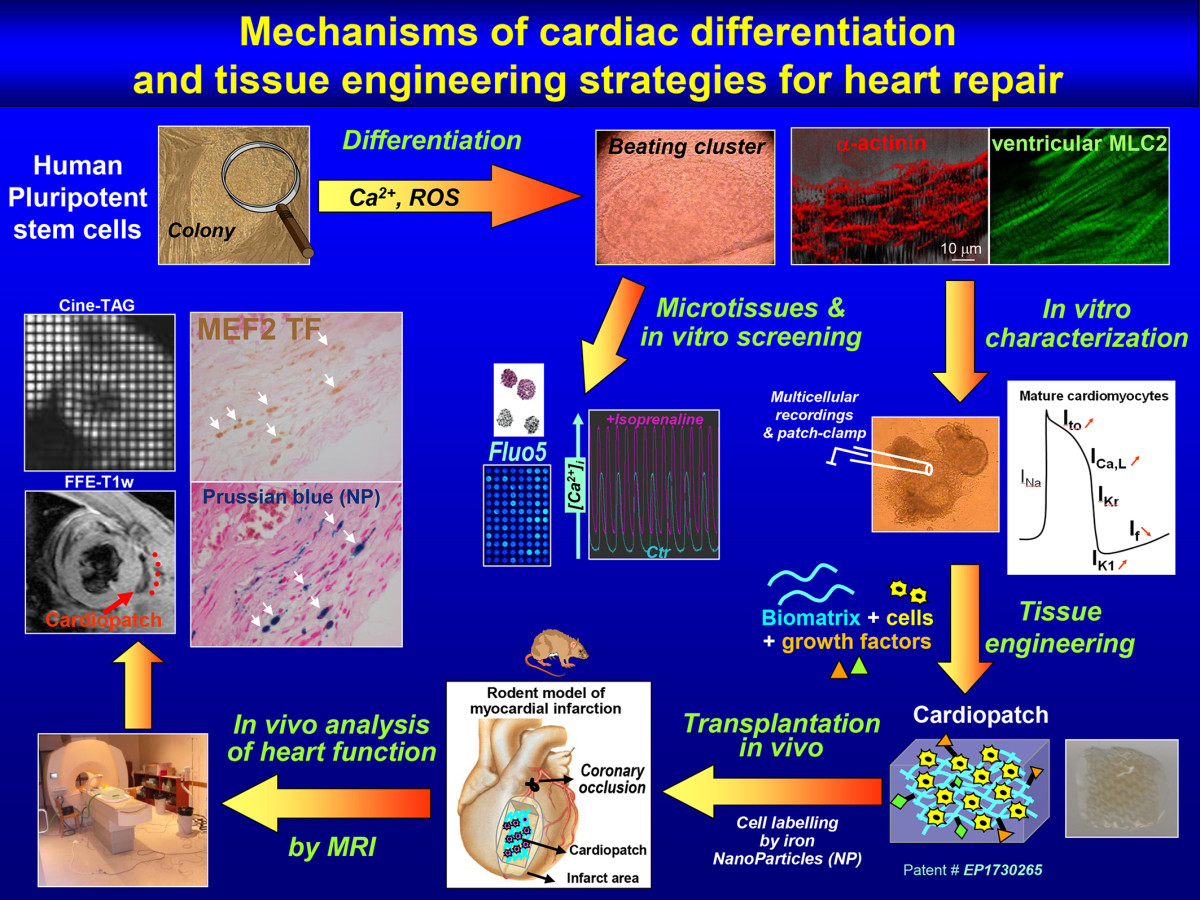
Cardiogenesis of human pluripotent stem cells and strategies of cell therapy
Our research group mainly investigates the mechanisms of cardiogenesis and cardiac cells. Specifically, we study cells featuring trisomy 2,1 with the aim of uncovering the differentiation of human pluripotent stem cells - either embryonic or induced pluripotent stem mechanisms at the basis of congenital heart defects in patients with Down syndrome. We also study the potential of cardiac and skeletal differentiation of mesoangioblasts, a subpopulation of perivascular stem cells with great potential for muscle regeneration, in particular of genetic diseases such as muscular dystrophy. Our long-term goal is translational, i.e. the set-up of muscle regeneration strategies, based on tissue engineering, that are easily transferrable to the clinical setting.
Apply now
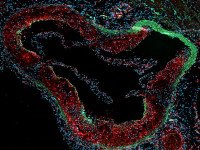
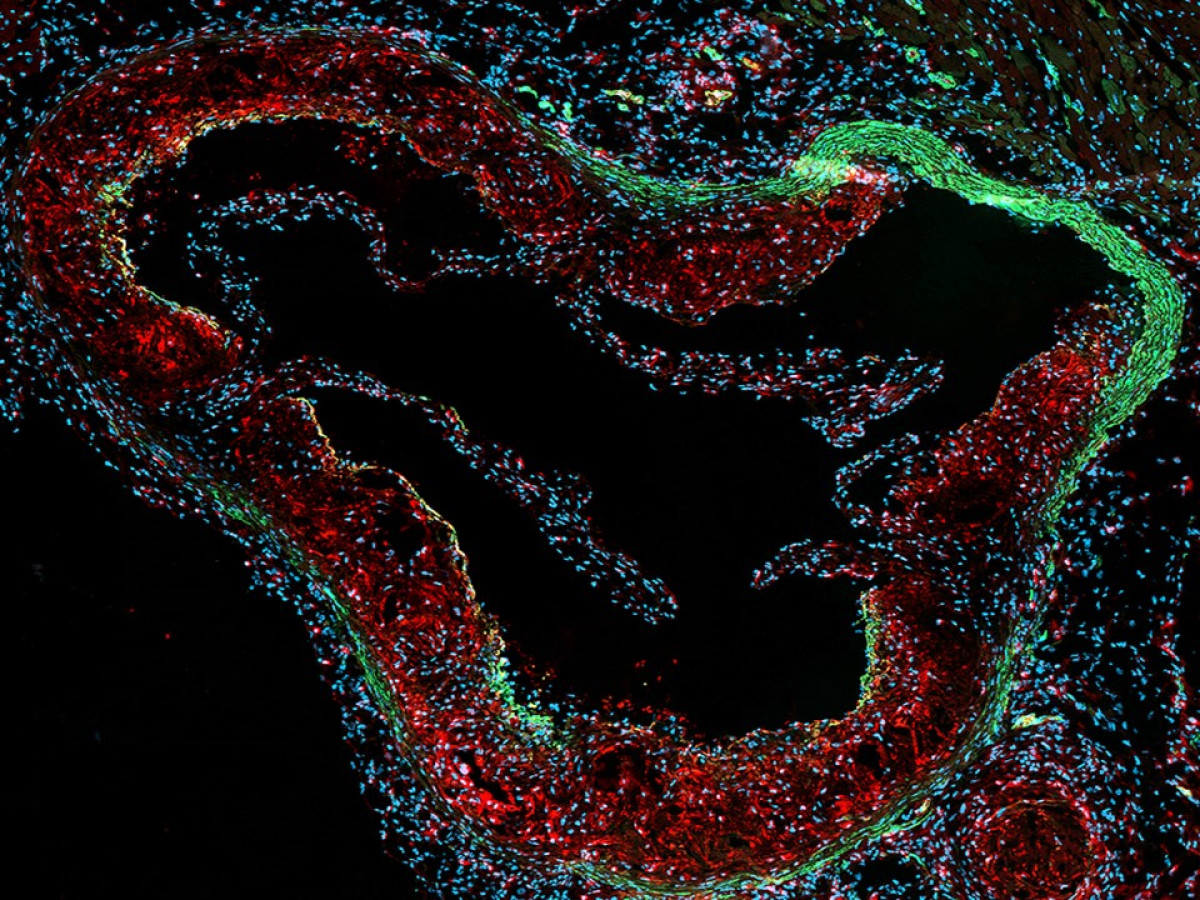
Smooth muscle cells in the pathophysiology of vascular disease
We have a longstanding experience in the implication of smooth muscle cell (SMC) heterogeneity in atherosclerosis. Using porcine coronary artery smooth muscle cells as in vitro model, we identified S100A4 as a marker of atherosclerotic SMCs, in both pig and human. Our recent results suggest that the extracellular form of S100A4 plays an unexpected role in SMC phenotypic transition associated with pro-inflammatory features. We are deciphering in vitro the signaling pathways involved in intra and extracellular S100A4-induced SMC phenotypic transition. In vivo, we study the role of S100A4 in the progression of atherosclerotic plaques in ApoE knockout mice. We are developing an atherosclerosis model with SMC tracing and SMC-specific deletion of S100A4 using the CRISPR-Cas9 approach.
Apply now

Circadian Endocrinology Lab
Circadian oscillation of biological processes has been described in most of the light-sensitive organisms on earth. It reflects the existence of underlying intrinsic biological clocks with near 24 h oscillation periods. There is an evidence for connection between metabolic pathologies and the circadian clockwork. The long-term goal of our laboratory is to identify the molecular basis of circadian rhythmicity in rodent and human peripheral tissues in physiological, and in obesity and type 2 diabetes conditions. We have setup the methodology for long-term recording of circadian reporter oscillations in human primary cells from metabolic tissues at population and single cell levels. In addition, we are interested in the connection between clock and cancer, in particular the thyroid cancer.
Apply now
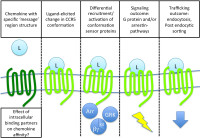
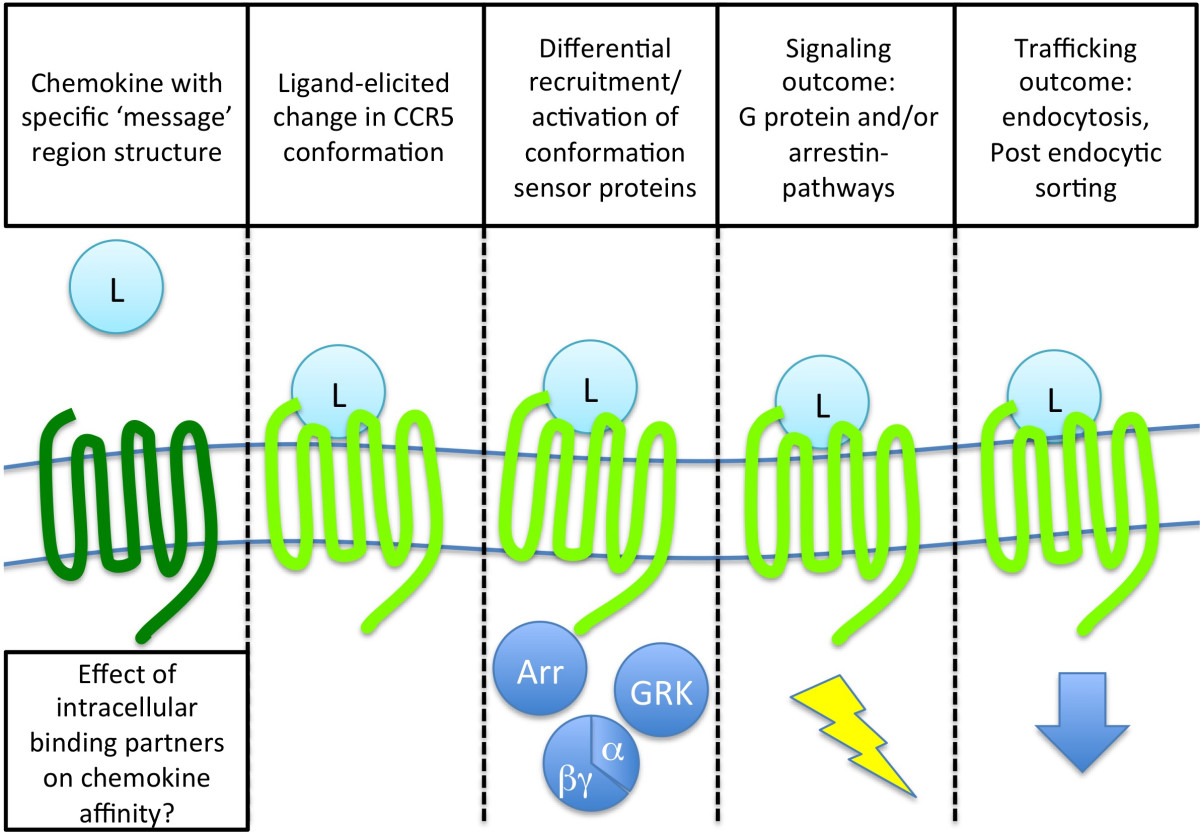
Studying CCR5 pharmacology with potent anti-HIV chemokines
CCR5, a chemokine receptor and a member of the GPCR superfamily, is the principal HIV entry coreceptor and a promising target for anti-HIV medicines. For over a decade we have been using a range of protein engineering approaches to discover and optimize analogs of the natural chemokine ligands of CCR5, culminating in the successful translation of our most promising chemokine analog into clinical trials. In parallel, our basic science research makes use of the chemokine analogs that we identified, together with innovative cell-based assays, as tools to study the biology and pharmacology of CCR5. Despite their structural similarity, these molecules show striking differences in anti-HIV potency, induction of receptor endocytosis and intracellular trafficking, and receptor signaling activity.
Apply now
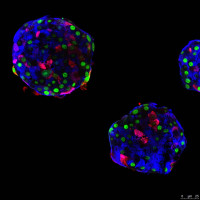
Regeneration of insulin cells by islet cell-type interconversion
We want to determine the origin of insulin-producing β-cells during embryonic development and in adults, whether during normal homeostasis or pancreatic regeneration after total β-cell ablation, using many transgenic mouse models and human pancreatic islets. Regeneration of new β-cells in the diabetic pancreas is the main research topic of the lab. We try to help devising innovative cell-based replacement therapies to treat diabetes. We have generated transgenic mice in which specific pancreatic cell types can be conditionally ablated. We have reported that β-cell loss in adult mice is followed by spontaneous β-cell regeneration, which mostly relies on the reprogramming of fully differentiated glucagon-producing α-cells into insulin producers.
Apply now

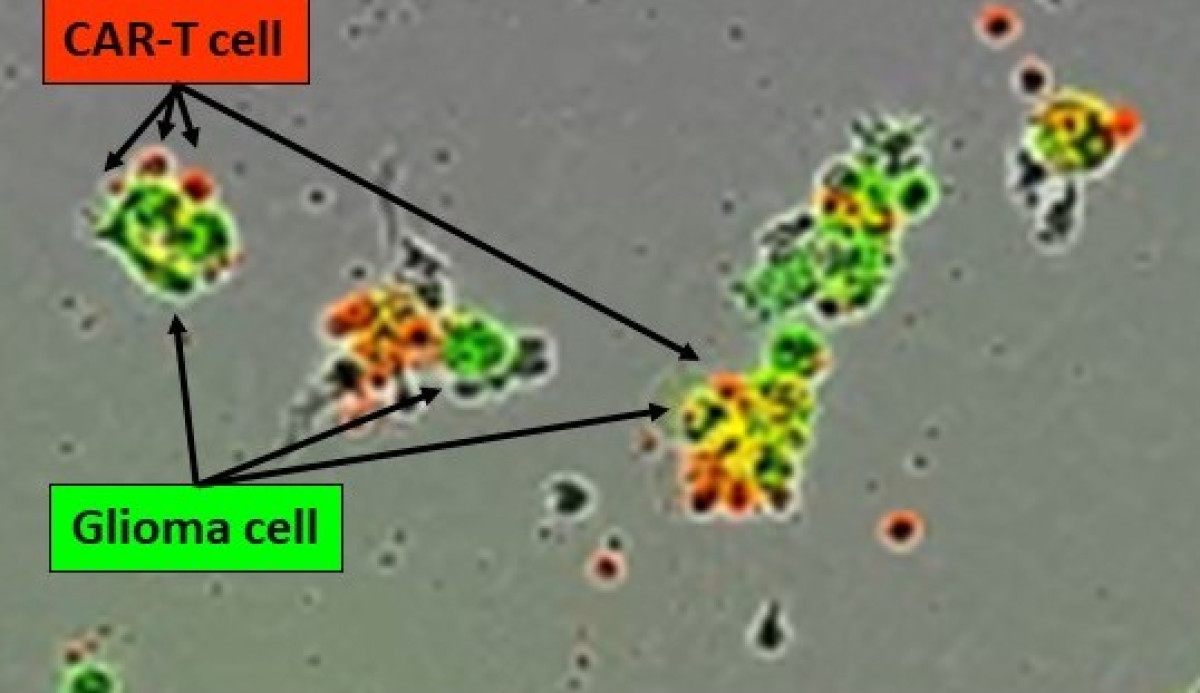
Brain Tumor and Immune Cell Engineering Laboratory
Tumors arising in the brain are notoriously hard to treat. We contributed to immune based therapies for gliomas, in the identification of cell surface markers of GBM in vivo and with the development multiepitopic vaccinations using synthetic peptides. We translated our research into the clinic in an early phase trial. We were able to demonstrate safety and immunogenicity. Our next endeavour is to develop innovative chimeric antigen receptor (CAR)-T cell therapies for GBM using multivalent approaches and targeting the immunosuppressive microenvironment. We also commit to mitigate neurotoxicity of FDA approved CAR-T cell therapies targeting B cell malignancies. We benefit from the generous support of the ISREC Foundation and are part of the Swiss Cancer Center Léman (SCCL).
Apply now
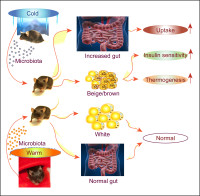
Obesity and insulin resistance
The main interests of our lab are the molecular mechanisms underlining metabolic diseases, primarily obesity and insulin resistance. The projects involve using state-of-art technologies and in vivo animal models, in vitro systems, cohorts of human patients, and lineage tracing studies, generally aiming at developing strategies for treatment of dyslipidemia, diabetes and obesity. Research interests include the roles of the adipose tissue and the gut in the regulation of metabolism; and the importance of the gut microbiota and the immune system in orchestrating the energy homeostasis. A deeper understanding of these axes is a prerequisite for optimizing therapeutic strategies to manipulate the gut microbiota and the host response to combat disease and improve health.
Apply now

LiRR: Laboratory for innovation in Radiobiology applied to radiotherapy
Radiotherapy (RT) is one of the most effective and cost-effective modalities for treating cancer and today more than half of all cancer patients will receive RT during their treatment. Thanks to technological and radiobiological improvements, RT is today a precise and conformational treatment. However, the treatment of radioresistant tumors remains limited by the tolerance of healthy tissues, which prevents increasing the RT dose and reaching a curative threshold. Thus, enhancing the differential effect of RT between tumor and healthy tissue, either by radiosensitizing the tumor or protecting healthy tissues selectively, remains the overarching goal in radio-oncology. Our laboratory develops translational research in radio-oncology to transfer advanced RT strategies to the clinic. This program aims at offering innovative treatments for our patients and strengthen multidisciplinary interactions amongst the fields of radiobiology, physics, adult and pediatric oncology, imaging, and pharmacology. FLASH Radiotherapy: Our group pioneered FLASH radiotherapy which is a breakthrough in radio-oncology based upon the administration of irradiation in an extremely short time (milliseconds or less) and at ultra-high dose rates (greater than 100 Gy/s), as opposed to standard conventional radiation which is delivered over several minutes at conventional dose rates (dose rates of 0.1 Gy/s). The main advantage of FLASH is to induce similar tumor control and reduce normal tissue complications compared to standard RT at conventional dose rate. While continuing our studies with protons and VHEE in collaboration with PSI and CERN, we are developing at HUG/UNIGE a new hub dedicated to X-rays for conformational FLASH radiotherapy. Our work primarily focuses on brain tumors, particularly rare pediatric tumors requiring special attention, and is funded by 7 academic grants. Personalized Radiotherapy: Focus on Breast Cancer Today, breast cancer is no longer considered a single disease but rather a set of different diseases. The understanding and classification of the disease at the molecular level have brought enormous benefits in overall survival and quality of life for patients, allowing for the personalization of treatment. Our goal is to personalize breast cancer radiotherapy based on its heterogeneity, which is considered today as the main challenge in innovation in breast radiotherapy.
Apply now
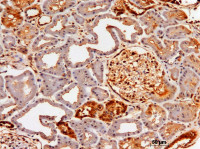
Pathophysiology of chronic kidney disease
The research focus of my group is to understand the pathophysiology of chronic kidney disease (CKD) and its complications. To this aim, we use animal models of kidney disease and proteinuria, but also patients samples including kidney biopsies and biological samples.Among current projects, the role of proteinuria on tubular cells is developped, linking proteinuria to fibrosis and cardiovascular complications. Modifications of oxygenation and tubular cell metabolism as factors of fibrosis progression are studied. Novel imaging techniques are developped in collaboration with the radiology department for early fibrosis detection. The goal of our translational approach is to determine new therapeutic pathways for CKD progression and complications, as well as novel diagnostic tools.
Apply now
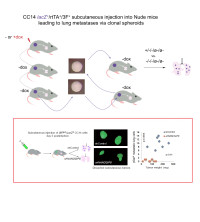
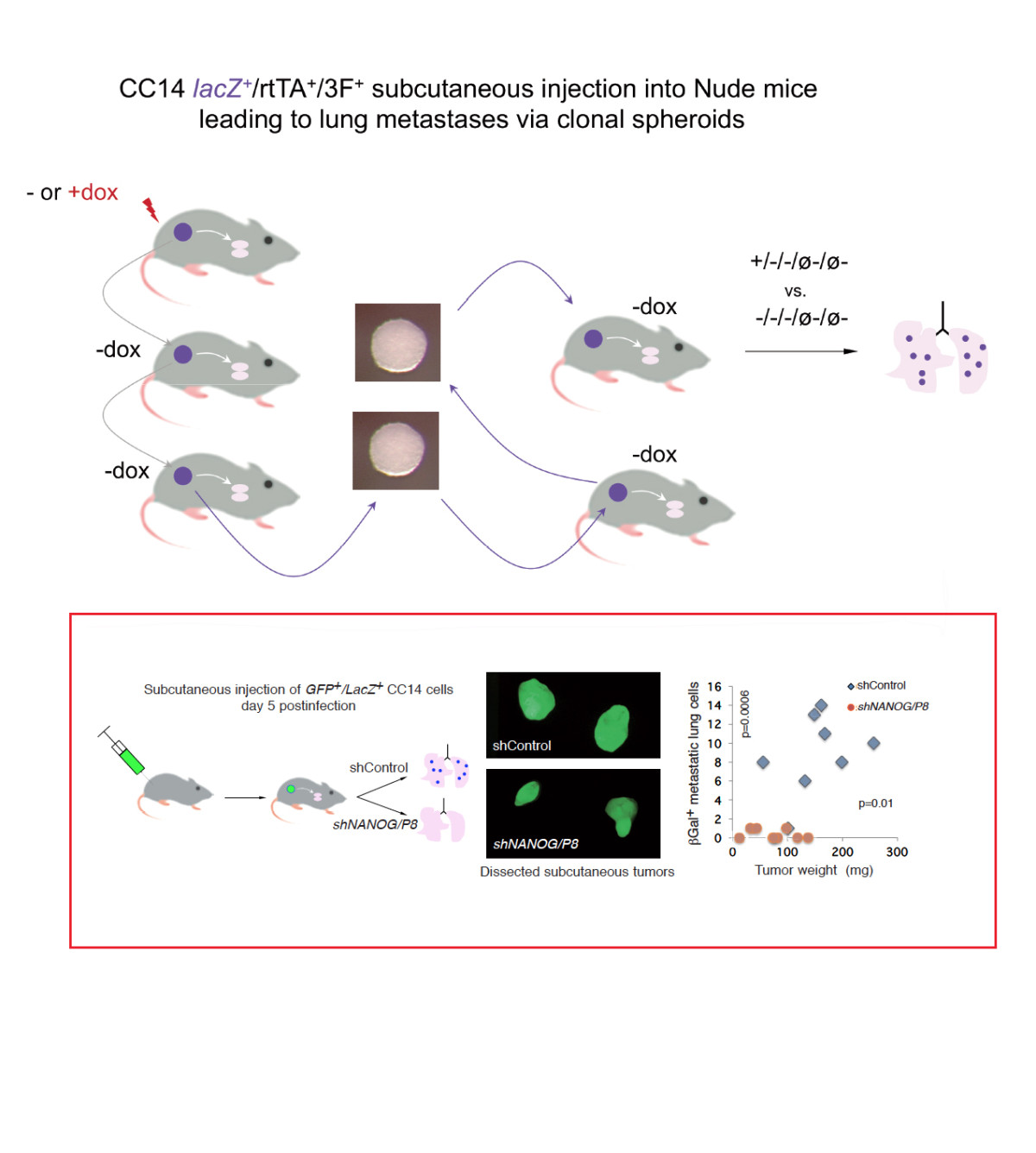
Cell plasticity and metastasis
Our overarching goal is to understand the rules of cell plasticity. Having worked extensively on early development, we now aim to understand how cells in primary tumors acquire metastatic fates. Currently the lab focuses on intercellular signaling and non-genetic changes that drive the acquisition of pro-metastatic states. We also explore the roles of embryonic signaling pathways in early steps of metastasis, investigating selected downstream targets and mediators.
Apply now
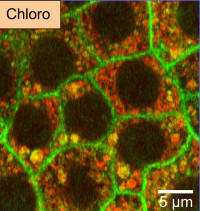
Physiology and pathophysiology of kidney tubule epithelial cells
We are interested in body fluid homeostasis with a focus on renal handling of water and electrolytes. We are studying the adaptation of renal tubular cell transport properties and metabolism to various amounts of dietary sodium and potassium ions; the coupling between transcellular and paracellular sodium transport and the effect of variations of sodium transport by renal tubular cells on primary cilium length and function. In addition, using several models of chronic kidney disease such as unilateral ureteral obstruction and inducible glomerular or tubular injury, we are currently assessing the role of the mineralocorticoid receptor and dietary sodium and potassium intakes in the progression of renal injury.
Apply now

Metagenomic mechanisms of infectious and non-communicable diseases
The Genomic Research Laboratory is actively involved in the development of methods in metagenomics and their application for medical research and diagnosis. Research interests: • Investigation of the mechanisms underlying Staphylococcus aureus biofilm formation, intracellular survival and antibiotic resistance • Development of rapid antimicrobial susceptibility testing (AST) methods
Apply now

Bioethics
Our research focuses on ethical issues in medicine and life sciences. Some of our projects explore philosophical concepts such as vulnerability or altruism. For example, our team published a definition of vulnerability enabling more precise protections, which was integrated into the Declaration of Helsinki and the CIOMS guidelines. Others projects look empirically at morally relevant aspects of care, research, or health policy. For example, we conducted studies demonstrating the reality of physician bedside rationing in Europe, the US, and Ethiopia, and showing that discussions should focus on how, rather than on whether, it was practiced. We also explore concrete issues from a normative perspective with a view to developing applicable and justified solutions.
Apply now

Chromosome segregation
Our group studies chromosome segregation in human cells. We aim to understand how during cell division the components of the mitotic spindle, such as centrosomes, kinetochores, and microtubules coordinate their function to establish a proper bipolar spindle and achieve faithful segregation of sister chromatids. Our interests focus on the fundamental molecular mechanisms controlling mitosis, how defects in these mechanisms lead to chromosome segregation errors in cancer cells, and how these errors can be exploited for anti-cancer treatments. Our studies rely on high-resolution quantitative light microscopy combined with chemical and genetic perturbations, cell biology, and biochemistry. Our group is part of the Translational Research Centre in Oncohematology.
Apply now

Cardiovascular and Hemodynamic Propulsion Research
The Charles Hahn Hemodynamic Propulsion Laboratory (LPHD) of the Cardiovascular Surgery Department is developing an integrative platform to study in vitro arterial circulation hemodynamics under physiological and pulsatile conditions replicating the cardiac cycle. Our research focuses on heart failure, structural heart disease, valvular heart disease, hemodynamics, flow modeling and the development of new therapeutic devices. We are also studying the function of endothelial cells in vessels used in cardiac surgery during coronary bypass surgery, using an organ bath. The aim is to gain a better understanding of the consequences for the patient's post-operative and long-term clinical course.
Apply now
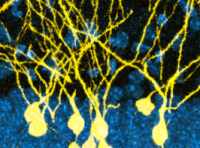
Epigenetic regulation of brain development
Our goal is to understand what drives brain cell diversity during development. We focus on the role of epigenetic regulators that control chromatin accessibility and gene expression. To edit the epigenome in living cells we recently developed a CRISPR-based technology called FIRE-Cas9 that allows for rapid and reversible recruitment of chromatin regulators to any genomic locus. The lab also uses mouse stem cell cultures, in vivo models and state-of-the-art genomics techniques to study mechanisms of chromatin regulation in the brain. Our aim is to gain novel insight into neurodevelopmental disorders which are frequently caused by mutations in epigenetic regulators.
Apply now

Bioengineering and Neuroregeneration
Traumatic and non-traumatic forms of injuries often result in tissue damage and disorders. Consequently, life-long disabilities represent a huge socioeconomic burden for the individual and on society. Tissue repair and regeneration is a complex and dynamic process requiring the exogenous factors regulating the multiple pathways, particularly in the case of neuronal tissues. For addressing these challenges, our group aims to advance translational research strategies by developing novel biomaterials, engineered hydrogels, and bioengineered cells and tissue grafts for modulation of the host-graft interaction and for regulating the tissue regeneration process.
Apply now
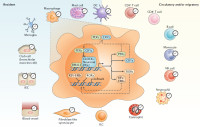
Circadian control of the immune system
We study circadian rhythms in the immune response using an interdisciplinary approach at the intersection of immunology, neuroscience and (patho)physiology. The recruitment of leukocytes to tissues and their localization within tissues plays a crucial role in the immune response. Recruitment of leukocytes to tissues underlies a circadian, i.e daily rhythm. This supports accumulating evidence for circadian oscillations in many components of the immune system. Specifically, the focus of our group is on how neural influences regulate the circadian migration of leukocytes to tissues, which promigratory factors control these rhythms and whether they can be altered by surgical, pharmacological or genetic interventions. The goal is to provide novel mechanistic insight into the systemic regulation
Apply now

Allogeneic Universal Chimeric Antigen Receptor (CAR) Invariant Natural Killer T (iNKT) Cells Against Acute Myeloid Leukemia
The use of Chimeric antigen receptor (CAR) T cells in acute myeloid leukemia (AML) faces major obstacles. Targeting antigens shared by AML cells and normal myeloid cells leads to bone marrow aplasia. Moreover, autologous T cells are often impaired after intensive chemotherapy. The overall objective of this project is to generate human invariant Natural Killer-T (iNKT) cells against one or several antigens expressed at AML surface. Our central hypothesis is that CAR iNKT cells will exert potent antitumor effects against AML cells through the combinatorial engagement of the CAR and the invariant TCR without inducing myelotoxicity and GvHD. Successful completion of this project will provide pre-clinical evidence for the use of CAR iNKT cells as a potential treatment of AML
Apply now

Cell Therapy & Musculoskeletal Disorders
Our research group is involved in basic and translational research in the field of osteoarthritis (OA), led by Pr Didier Hannouche, and in the field of skeletal muscle stem cells (MuSC), led by Dr Thomas Laumonier. In OA, articular cartilage follows a progressive degeneration, where low-grade inflammation could play a pivotal role through an oxidative stress-dependent mechanism and exposure to reactive oxygen species (ROS). We hypothesize that ROS production by chondrocytes through NADPH isoform 4 (NOX4), plays a noxious role in OA pathogenesis. For this purpose, we investigate the implication of NOX4 in the pathophysiology of OA, both in vivo and in vitro. The laboratory has also a long experience in the isolation, culture, characterization, and transplantation of human MuSC. The research work focuses mainly on the development of cell therapy strategies in the treatment of skeletal muscle disorders, including myopathies. We demonstrated that human muscle reserve cells (MuRC), generated in vitro, are like quiescent MuSC with properties required for their use in cell therapy. The objective of the ongoing studies is to characterize the molecular signature, the metabolic profile of human MuRC and to identify new mechanisms that regulate the function of these cells in vitro and in vivo.
Apply now

MASLD/MASH and insulin resistance
Our laboratory focuses mainly on the link between insulin resistance, a major feature of type 2 diabetes and obesity, and metabolic dysfunction-associated steatotic liver disease (MASLD)/metabolic dysfunction-associated steatohepatitis (MASH). The topic is particularly important because the prevalence of type 2 diabetes is constantly increasing and could affect 600 million people worldwide in the next decade. MASLD is now the most common chronic liver disease in the world, affecting nearly a quarter of the general population and up to 70% of people with type 2 diabetes. Going unnoticed for a long time, MASLD can lead to complications such as liver fibrosis, portal hypertension, cirrhosis and even hepatocellular carcinoma (liver cancer). It is therefore essential to identify patients with MASLD/MASH early, to determine which patients are likely to have an unfavorable progression and to find therapeutic targets to prevent and cure MASLD/MASH. With this in mind, our laboratory carries out translational research, starting from the deepening of fundamental knowledge through the use of cell culture and rodent models, to arrive at new applications beneficial to patients. The therapeutic targets most investigated within the laboratory concern the signaling pathways of FGF21, GLP-1 receptor agonists and cytokines of the interleukin-1 family. Our research also focuses on the role of the intestinal microbiota and the enterohepatic axis in the occurrence and progression of MASLD/MASH.
Apply now

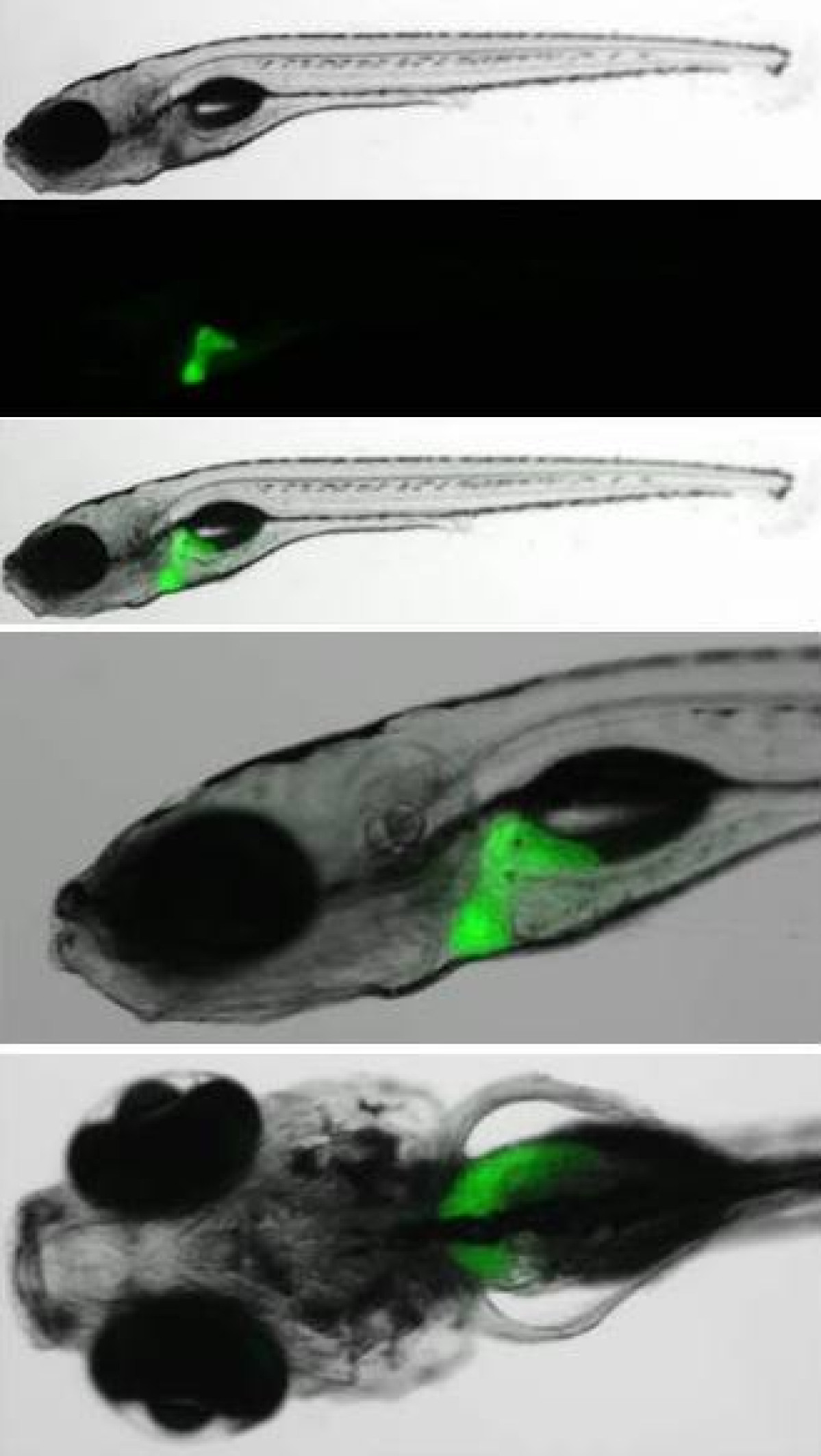
Hemato-vascular development in the vertebrate embryo
The blood tissue consists of cells with various roles, involved in oxygenation of tissues or immunity. In mammals, these cells are constantly replenished in the bone marrow, from hematopoietic stem cells (HSCs). HSCs are multipotent and can self-renew. They derive from a small subset of HSCs produced during embryogenesis. Our main goal is to understand the molecular cues involved in HSC specification, expansion and differentiation. We use the zebrafish as a model to study HSC biology. Fertilization occurs externally, producing large number of embryos that can be easily manipulated. Importantly, zebrafish blood cells, as well as the molecular pathway necessary to their differentiation are conserved with other vertebrates therefore allowing us to translate our findings to the human model.
Apply now
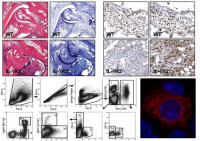
IL-1 cytokines in inflammation and immune responses
My laboratory is interested in the role of cytokines of the interleukin (IL)-1 family in the immune and inflammatory responses. IL-1 is a cytokine that is critical in the innate immune response to different types of endogenous or exogenous noxious agents. In addition, other IL-1 cytokines such as IL-18, IL-33 and IL-36 share structural characteristics, bind to the same family of receptors, and stimulate similar intracellular signals as IL-1. Interestingly, natural inhibitors, including receptor antagonists and decoy receptors exist for all the IL-1 cytokines. Our research group is generating and working with different genetically modified mouse lines, to elucidate the role of these cytokines in several models of inflammatory diseases.
Apply now
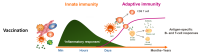
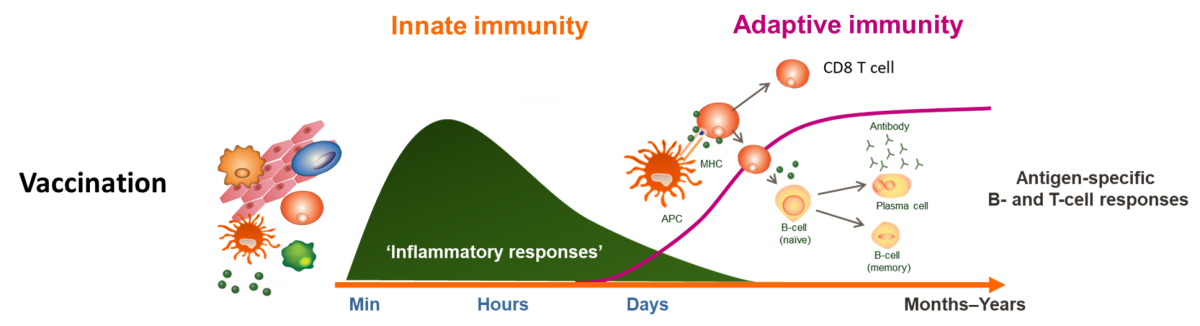
Role of innate immunity in vaccine response
The ability of a vaccine to induce a long-lasting memory response is conditioned by the initial stimulation of the innate immune system, in particular antigen-presenting cells. Individuals with immunocompromising conditions, such as transplant or patients or those taking immunomodulatory drugs, are generally less responsive to vaccines.Through a translational approach, combining clinical research studies and studies in animal models, we aim at defining which alterations in innate immune response to vaccines (changes in gene expression level, inflammatory mediators..) due to the disease or drugs translate into a change in the quality of the antigen specific antibodies, T and B cell response. In addition, we explore the mechanism of action of vaccines in animal models.
Apply now
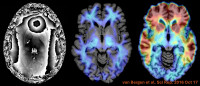
Personalized brain health
One of our major research goals is to identify biological and environmental determinants of maintaining mental health in old age. Our research aims to advance individualised precision medicine interventions for the treatment of age-related neurodegenerative brain disorders such as Alzheimer's disease (AD). We are particularly interested in characterising altered central nervous system function associated with progressive cognitive dysfunction, depression, anxiety, apathy and difficulties in social interaction. To this end, a multidisciplinary longitudinal research cohort (Geneva Geriatric Mental Health Inventory) has been established. Complex clinical assessment is combined with state-of-the-art neuroscientific methodology. These include advanced high field neuroimaging studies, innovative molecular radiopharmaceutical imaging, blood biomarker and genomic studies. Several ongoing research collaborations have been established with basic research groups at HUG, Campus biotech Geneva and Lausanne, ETH and University of Zurich, Basel Neurology and abroad.
Apply now
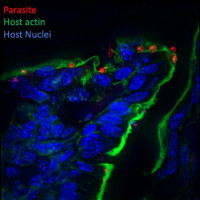
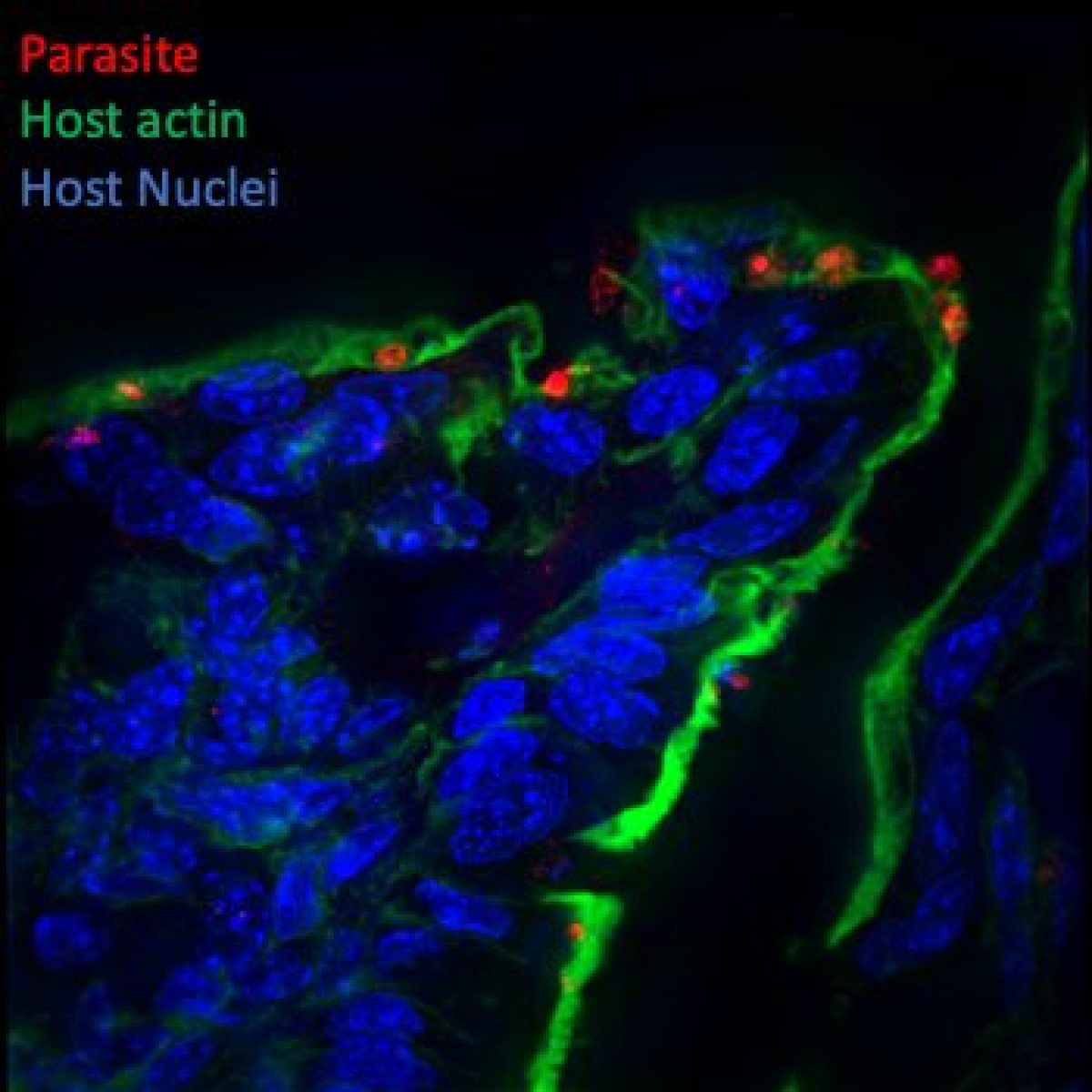
Identification and characterization of Cryptosporidium secreted effectors hijacking host cellular functions
Cryptosporidium is a leading cause of diarrheal diseases and death in infants, and no vaccine or efficient drugs are available. The parasite is an obligatory intracellular pathogen that infects epithelial cells of the intestine and resides inside a vacuole. It hijacks the host cell to its own advantage notably via the formation of a host actin pedestal and a complex interface. Additional modifications of the host cell include the remodeling of membranes during invasion, the destabilization of epithelial cell junctions and the shortening of microvilli that contribute to the diarrheal symptoms. Subverting host cell functions is essential for Cryptosporidium invasion and survival but poorly characterized. Understanding how the parasite achieves it is the main goal of this research.
Apply now
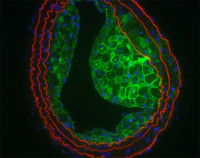
Connexins and pannexins in cardiovascular disease
Connexins (Cxs) and pannexins (Panxs) form transmembrane channels that control intercellular communication and contribute to vascular homeostasis. Mutations or polymorphisms leading to channel dysfunction have been implicated in several human vascular pathologies, including atherosclerosis, thrombosis and lymphedema. Our research group is interested in the mechanisms by which Cxs and Panxs participate in cardiovascular physiology and disease. In particular, the lab focuses on the transcriptional regulation of Cxs and Panxs in response to wall shear stress, the frictional drag force of the flowing blood on the vascular wall. Moreover, we are studying the Cx and Panx interactome, identifying novel protein partners and their role in vascular (patho)physiology.
Apply now

Identification of key viral and host factors modulating rhinovirus and enterovirus pathogenicity
Rhinoviruses (RV) and enteroviruses (EV) are leading causes of infections in humans. Although closely related at a genetic level, they are characterized by an important genetic variability, illustrated by the existence of more than 250 different types. This genetic heterogeneity is paralleled by an important phenotypic diversity. RV infection is restricted to the respiratory tract, whereas EVs can cause viremia, spread to multiple sites, and have been associated with over 20 distinct syndromes, from common cold to encephalitis. Our research explores the pathogenic diversity of RV and EV. Using molecular, cellular and biochemical tools, we aim to determine and characterize the genetic factors that underlie clinically relevant phenotypic traits, such as virulence and neurotropism.
Apply now
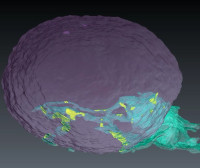
Mechanisms of antigen cross-presentation
Antigen cross-presentation by dendritic cells (DCs) is paramount for immunity against intracellular pathogens and cancer. Immunotherapies are promising strategies that at least in part rely on cross-presentation. However, many present formulations are poorly efficient, in part because the mechanisms that govern cross-presentation are not fully understood. Endoplasmic reticulum (ER) proteins are required, but the precise role of the ER has remained elusive. We investigate novel ER structures called membrane contact sites, aiming not only to clarify how the ER participates in regulating cross-presentation, but also to identify novel pathways that may be exploited to improve cross-presentation efficiency in formulations of DC-based immunotherapies.
Apply now
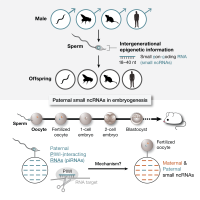
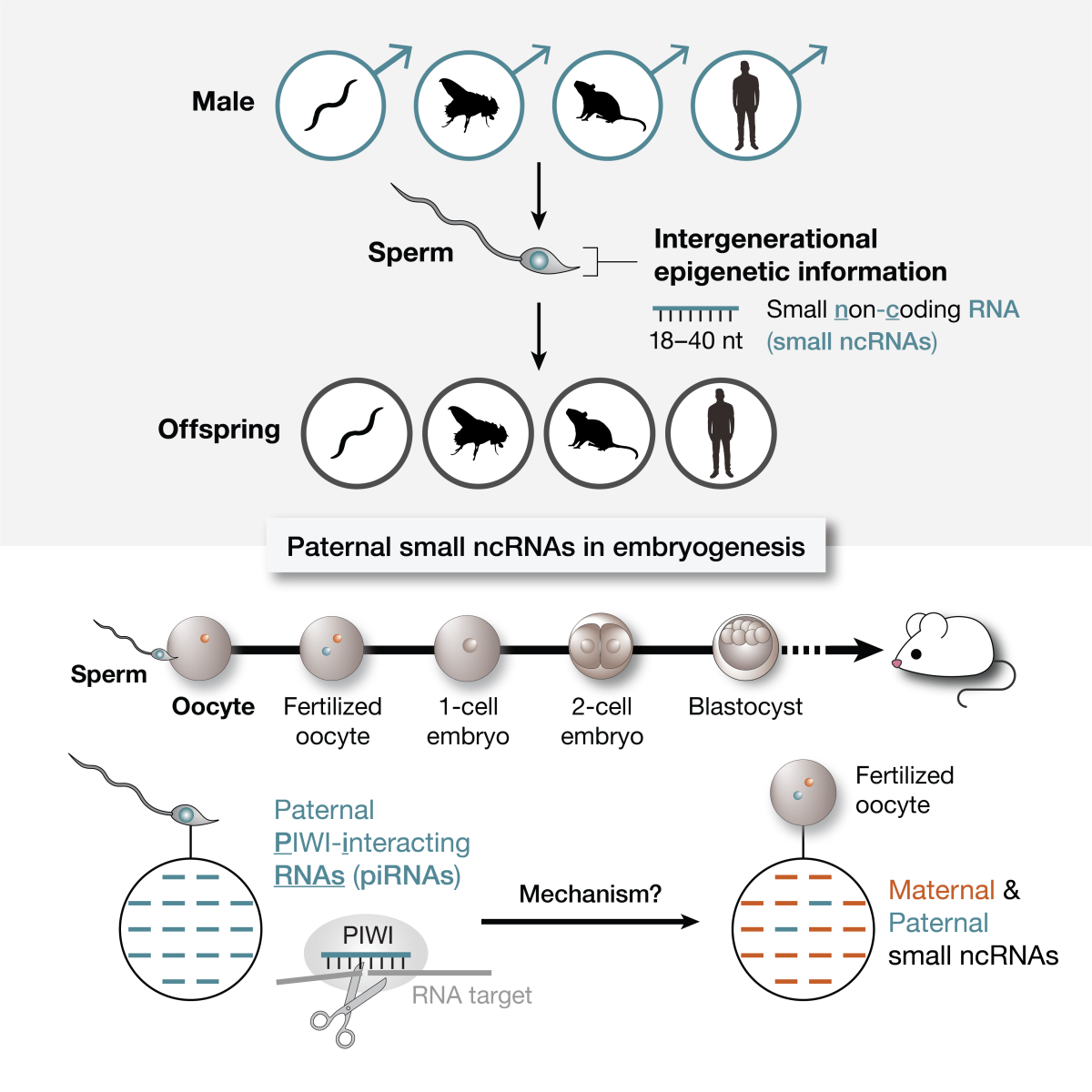
Defining the role of paternal PIWI-interacting RNAs in early embryo development
Unlike maternal contribution, how fathers influence offspring development is poorly understood. Evidence has brought small RNAs to light as crucial paternal regulators, but the mechanism is unknown. Our work suggests a paternal role for germline-enriched small RNAs—PIWI-interacting RNAs (piRNAs)—in embryo development. Hallmark molecular events in a newly formed zygotic embryo, such as activation of the zygotic genome, determine its viability. We aim to understand the role of paternal piRNAs in such key events by interrogating dynamics of mRNAs, epigenetic marks, and small RNAs in embryos using high-throughput sequencing, CRISPR-mutant mouse models, and time-lapse imaging. Using piRNA as a molecular framework, our goal is to illuminate how paternal small RNAs influence embryo viability.
Apply now
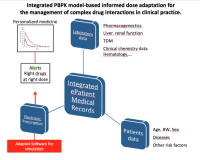
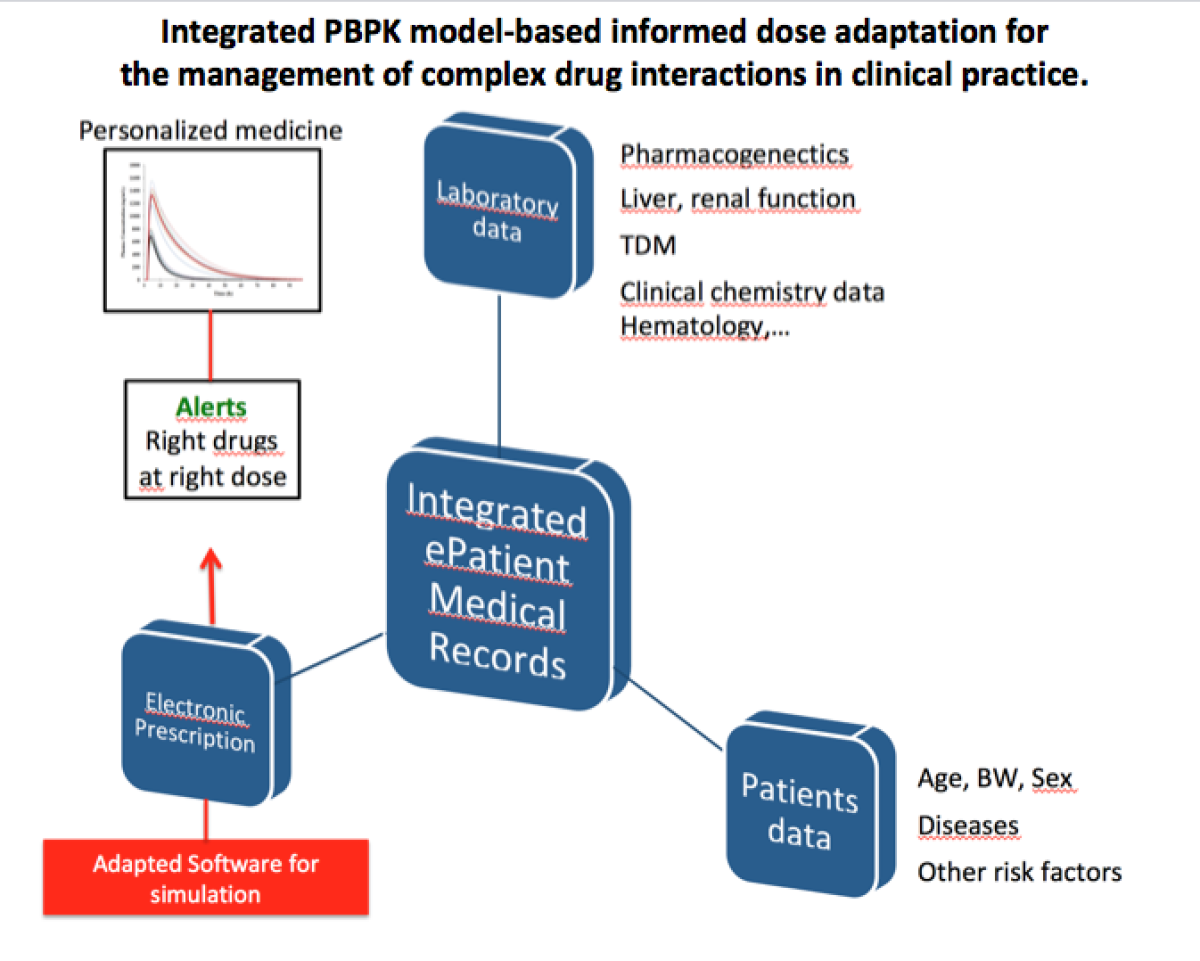
Translational Research in Drug Metabolism and Transport and Physiologically Based Pharmacokinetics use in clinical Setting
Physiologically Based Pharmacokinetics modeling (PBPK) use in clinical setting has become an important challenge to help clinicians tailoring medication based on genetic, environmental as well as patients’ characteristics. Drug-drug interactions (DDIs) are of particular clinical interest because of their high prevalence among polymorbid patients and their potential deleterious clinical implications. DDI prediction represents one of the most promising domains for a successful use of PBPK. Drug metabolic enzymes and transporters are the most important factors in interindividual variability. Several in-vitro models are used to generate data evaluating metabolism and transport of drugs. These data as well as in-vivo data are incorporated in the software for an optimal medication adjustment.
Apply now

Pathogenesis of Biofilm-Associated Infections
Biofilms contribute to the antibiotic and immune tolerance of a variety of bacterial infections, including common urinary tract and wound infections, rendering them difficult to treat. Biofilms that are polymicrobial in nature can display heightened antibiotic tolerance, amplify disease severity, and further complicate treatment options. Our research aims to better understand the host-pathogen interactions of polymicrobial infections so that we can identify improved treatment strategies in the future. Using genetic and genomic approaches, coupled with in vitro and in vivo models of host-associated biofilm formation and persistence, we study the bacterial and host factors that impact interspecies interactions between co-infecting bacteria and the mammalian host.
Apply now

Epigenetic Control of Developmental Processes
Our aim is to understand how epigenetic information, in particular the 3D organization of human and mouse genomes instruct the development of organs and structures during embryogenesis. To map and functionally disrupt these chromatin states, we employ a variety of state-of-the-art technologies in pluripotent stem cells and in vivo during embryogenesis. The understanding of these basic concepts is critical to unravel the molecular mechanisms that underlie pathological gene misregulation in congenital malformations and cancer as well the mechanism that enable evolutionary novelties and diversity of life.
Apply now

Unité de recherche clinique en onco-hématologie
L’unité de recherche clinique en onco-hématologie a pour but de donner accès, dans le cadre d’essais cliniques, à de nouvelles thérapeutiques aux patients adultes souffrant d’affections onco-hématologiques. Actuellement 45 études sont ouvertes. La majorité des traitements sont des immunothérapies et/ou des thérapies ciblées, c’est-à-dire des molécules reconnaissant des protéines situées à la surface ou à l’intérieure des cellules tumorales. Nous avons également des études évaluant l’impact d’autres interventions comme le mindfulness ou l’activité physique. Les études cliniques sont principalement des études de phases précoces c’est-à-dire des études de phase I ou II, permettant de déterminer le profil de sécurité et l’efficacité des traitements.
Apply now