Our groups
The doctoral program of the School of Pharmaceutical Sciences provides an extremely stimulating, international environment for motivated and outstanding young scientists offering first class opportunities and infrastructure/facilities to match. Research spans the pharmaceutical sciences – from fundamental and translational aspects covering molecular pharmacology, drug discovery including bioactive natural compounds, biopharmacy, drug delivery, pharmaceutical technology, pharmaceutical analytics through to clinical, community and hospital pharmacy – truly going from the bench to the patient. The doctoral program has 100-120 PhD students who are actively encouraged to present at (inter)national meetings, improve scientific/soft skills and build their professional network for their future careers.
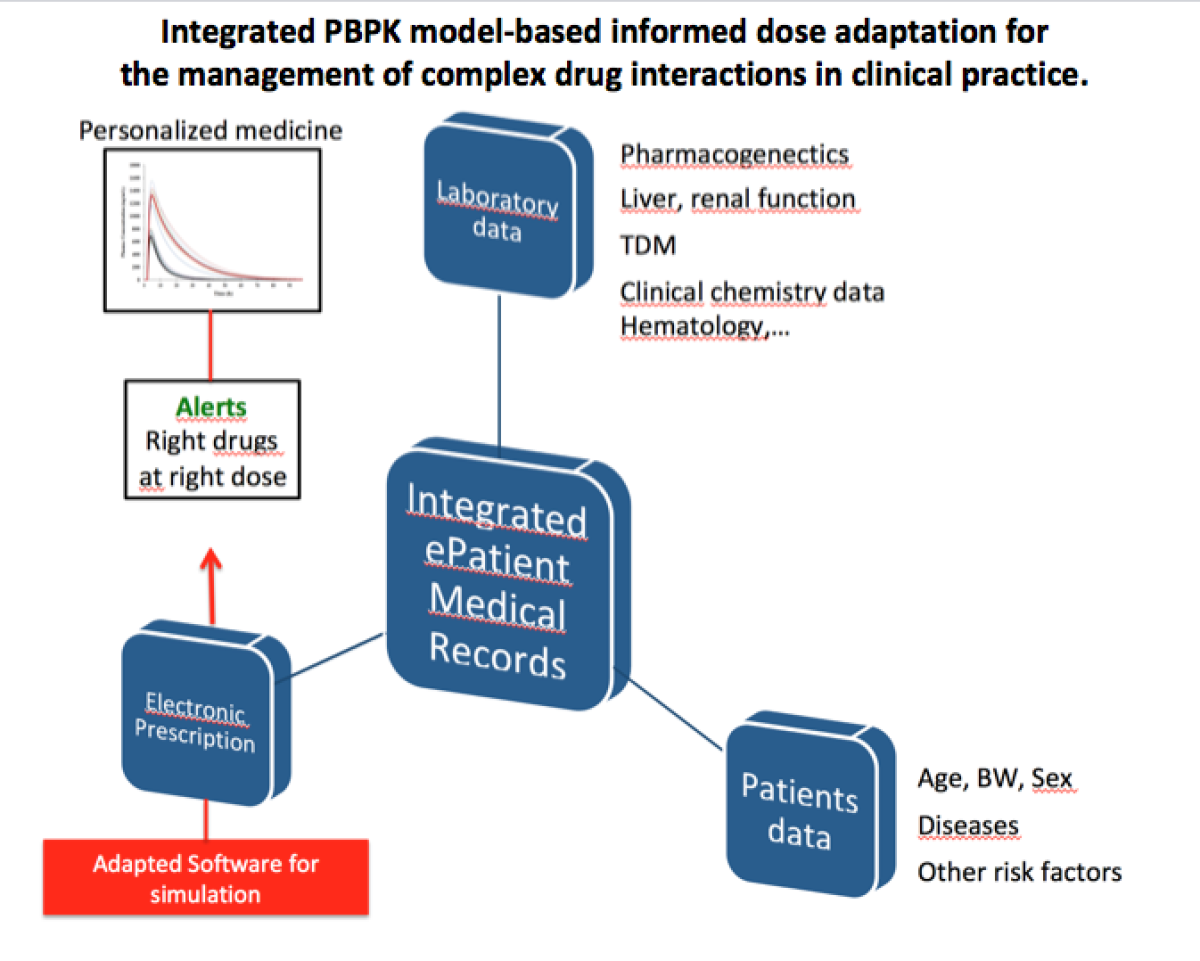
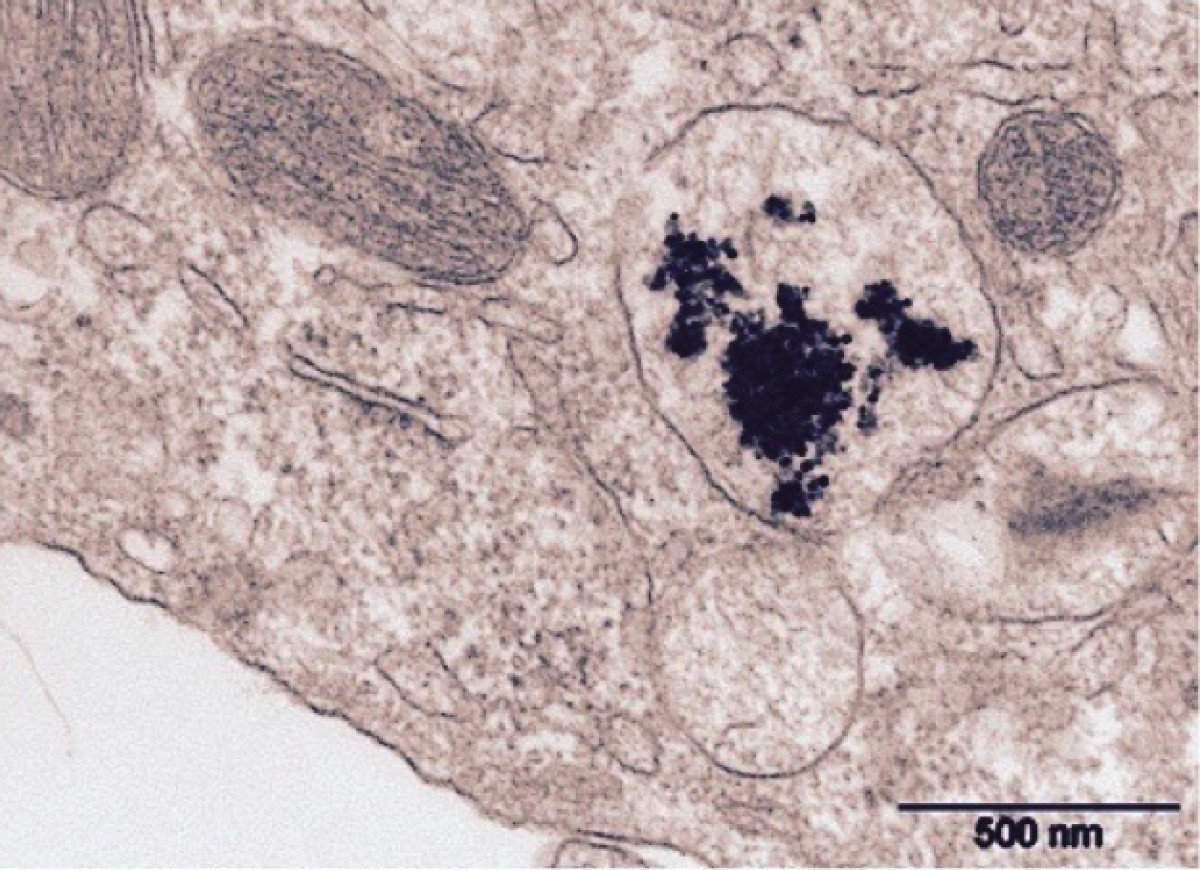
Personalized medicine: Managing interindividual variability in drug metabolism and transport
Interindividual variability in drug response is a major problem in patient care. The origin is multifactorial including intrinsic factors (genetic polymorphisms, diseases, age) or extrinsic factors (drug-drug interactions (DDI), toxics exposure). Drug metabolism and transport are responsible of a large fraction of the pharmacokinetic variability. Identifying these variables at the individual level permit to offer a personalized care reducing toxicity or inefficacy of drugs. Physiologically Based Pharmacokinetics modeling (PBPK) use in clinical setting has become an important challenge to help clinicians tailoring medication based on genetic, environmental as well as patients’ characteristics. PBPK approach can be used in vulnerable populations, for DDI evaluation and pharmacogenetics.
Apply now

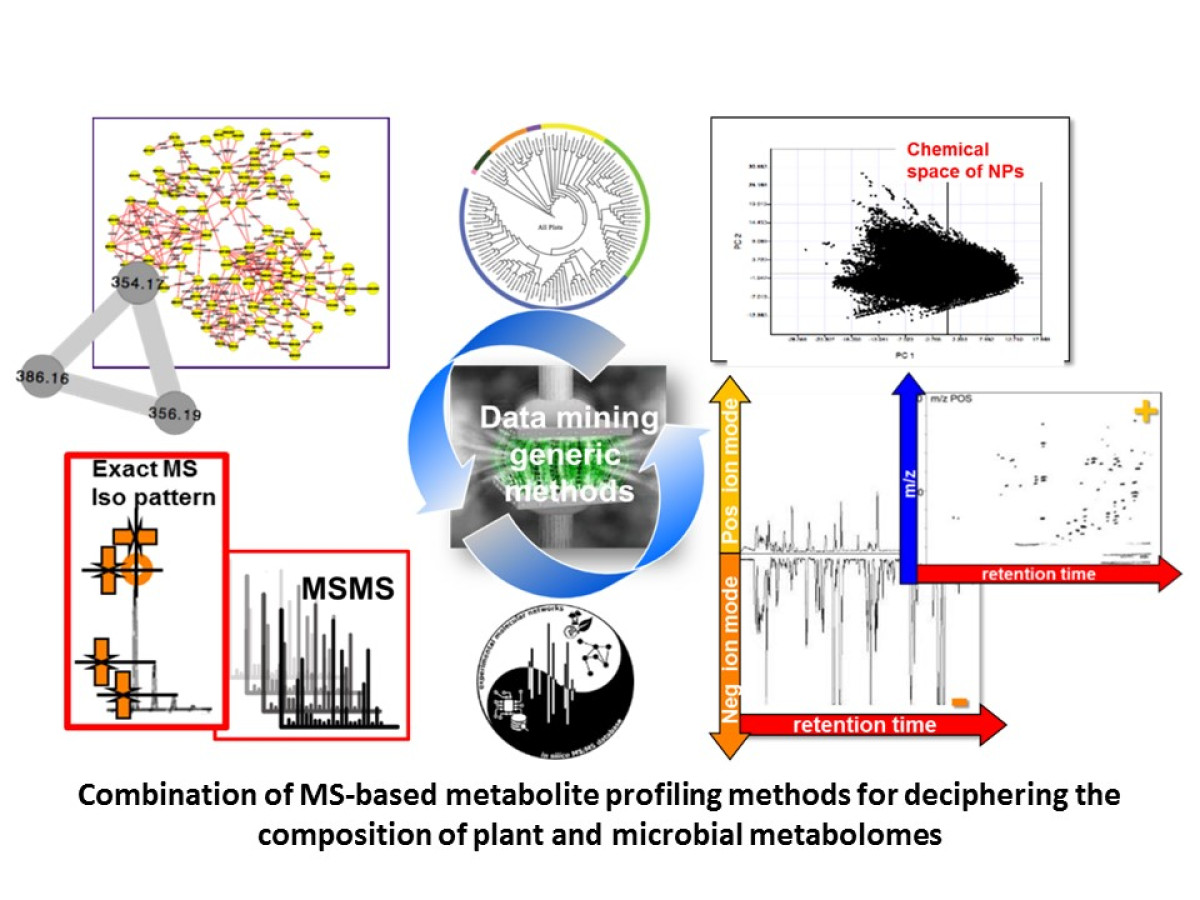
Biological Data Mining
Due to the ever-increasing number of signals that can be measured by modern analytical platforms, life sciences datasets not only become larger in size, but also more intricate in their structures. This gives rise to new challenges, and dedicated algorithms able to cope with their inherent properties are mandatory to extract the biochemical information. In that context, we develop data mining strategies, including chemometrics, machine learning and bioinformatics for the analysis of megavariate data. These tools aim at an efficient investigation of specific biochemical signatures in complex experimental designs, to combine multiple data sources through data fusion strategies, and incorporate biological knowledge into modelling workflows using efficient metabolite annotation.
Apply now
Investigation of bioactive natural products
Our group develops innovative MS- and NMR-based metabolomics strategies for natural products (NPs) research projects related to phytochemistry, microbial interactions and phytotherapy. We are specialised in the de novo structure identification of bioactive biomarkers at the microgram scale using a miniaturised approach that combines activity-based HPLC profiling and high content bioassays such as zebrafish. Our main research focuses on the search of novel inducible bioactive NPs in response to biotic and abiotic stimuli as well as for the study of the mode of action of phytopharmaceuticals from a systems biology perspective. We have currently projects related to antimicrobials, epilepsy, cancer, diabetes, obesity, NP bioavailability, plant wound signalling, microbial quorum sensing.
Apply now
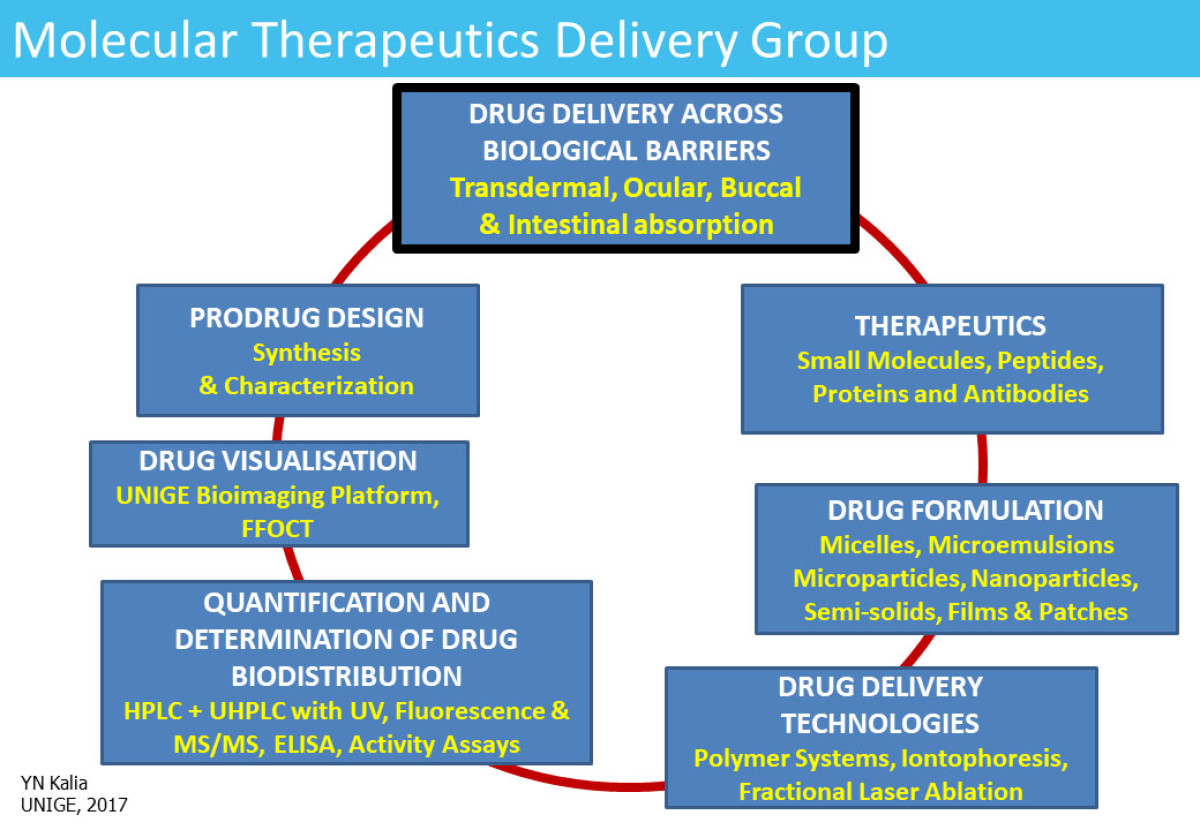
Molecular Therapeutics Delivery
Our research focuses on prodrug approaches and the development of formulation (e.g. micelles) and technology-based strategies (e.g. iontophoresis/fractional laser ablation) to improve drug delivery into and across biological membranes. We work with small molecules, peptides, and proteins (including mAbs). Our main area of interest has been cutaneous and transdermal delivery but in recent years we have applied our expertise to other routes of drug administration. There are ongoing projects in buccal and ocular (intracorneal/transscleral) drug delivery and the development of models for intestinal absorption. In addition to improving drug bioavailability, we focus on understanding drug biodistribution in the target tissue both quantitatively and through state-of-the-art imaging techniques.
Apply now
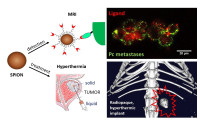
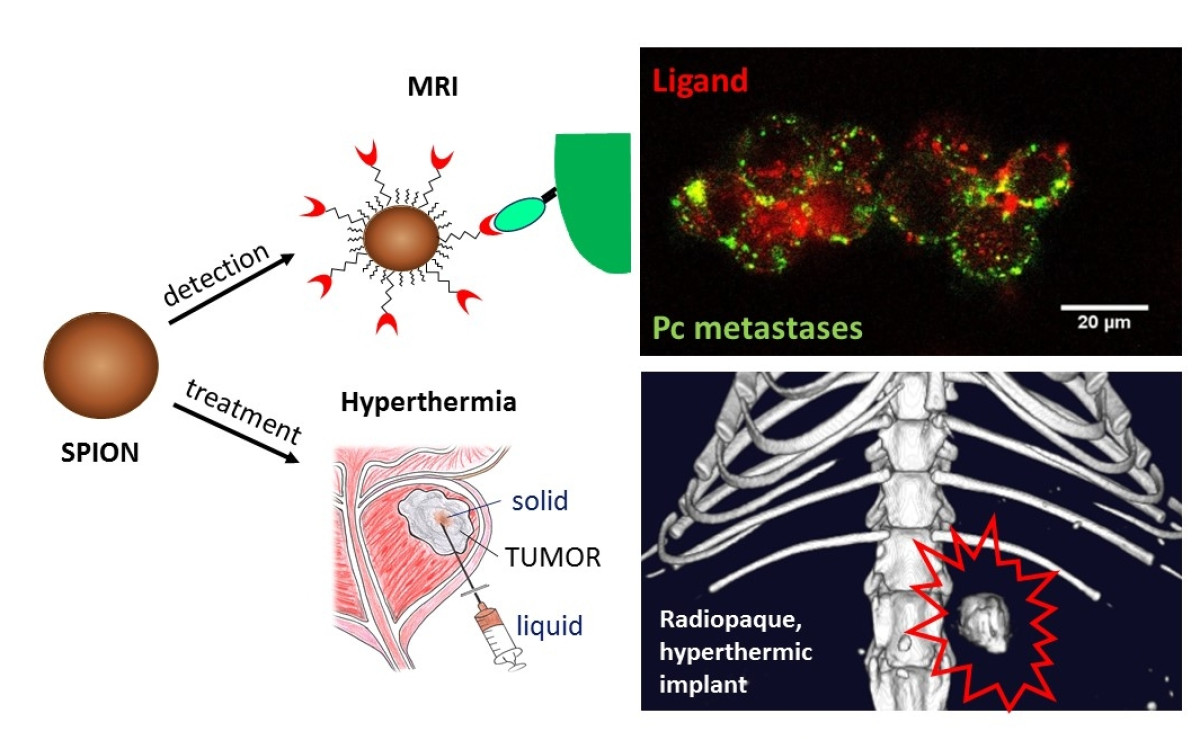
Advanced drug delivery carriers
Our research focuses on advanced drug delivery carriers, mostly nanocarriers or microparticles, for diagnostic and treatment. For instance, we designed microparticles releasing antiangiogenic tyrosine kinase inhibitors for the embolization of hepatic carcinoma, which lead to clinical developments. Aptamer-functionalized iron oxide nanoparticles targeting PSMA+ cancer cells were developed for metastasis imaging and subsequent hyperthermia treatment (Fig). Specific indications might also benefit from a sustained drug delivery matching the slow progression of the disease. We made significant advances in the fields of osteoarthritis treatment with chondroprotective and anti-inflammatory agents, or in the prevention of grafted vessel restenosis by long-term release of statins.
Apply now
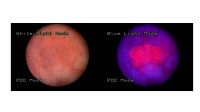
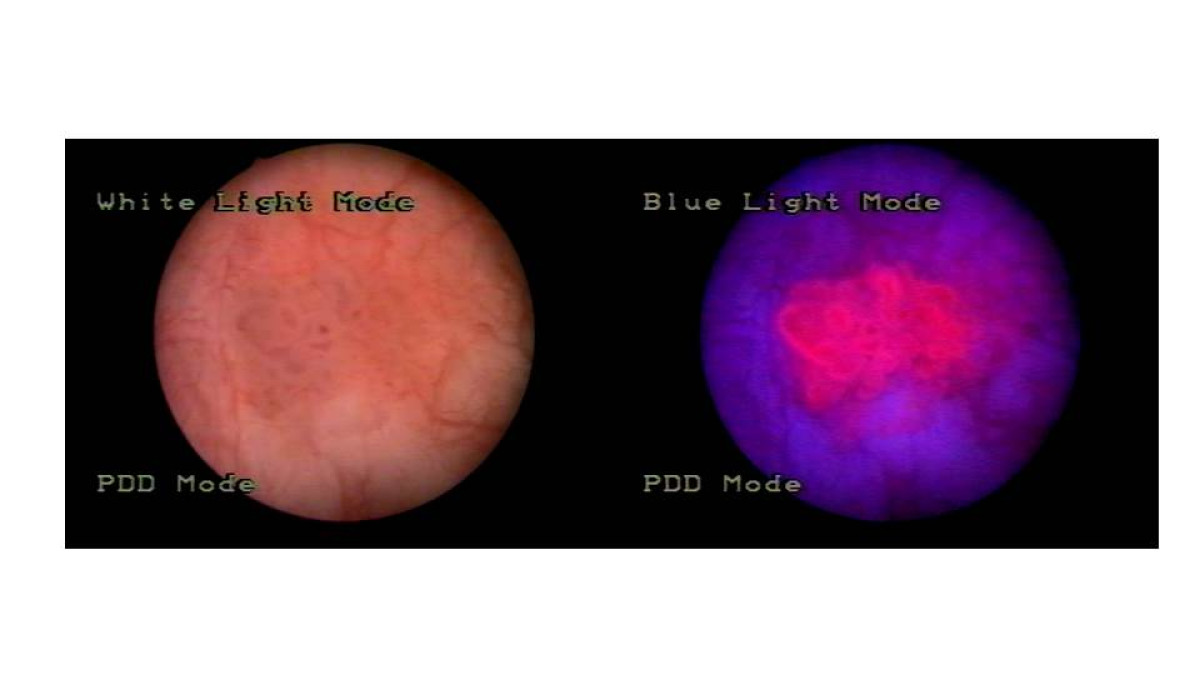
Translational research in Theranostics
Pharmaceutical sciences have evolved from the formulation of active principles to improve absorption and bioavailability to the design of drugs and therapeutic systems targeting specific molecular sites of disease. In our translational research we are working in collaboration with clinicians and biologist in diverse areas. We are developing new compounds for therapeutic and diagnostic purposes, especially on prodrugs that target altered enzymatic activities. For example we have developed inactivated prodrugs that release the active principle in presence of proteases overexpressed in tumors. In another research area, we have developed a new class of 5-aminolevulinc acid derivatives appropriate for systemic administration to score and destroy tumors with alterations in heme biosynthesis.
Apply now

Biomolecular and Pharmaceutical Modelling
The biomolecular and pharmaceutical modeling group, led by Prof. Francesco L Gervasio, focusses on the development of methods for biomolecular simulations with emphasis on enhanced sampling methods, as well as multiscale and coarse-grained models. The group crucially contributed to the development of methods for overcoming the timescale problem (metadynamics, parallel-tempering metadynamics, path collective variables, SWISH), which are widely used across different fields ranging from drug design to nanotechnology. We apply these methods to study a multitude of complex biomolecular phenomena, including protein dynamics and folding, ligand binding, allosteric mechanisms, and modes of action of cancer-causing mutations. Our simulations have guided the design of several allosteric drugs.
Apply now

Medication adherence and interprofessionality
The research plan of the unit aims at achieving a better understanding of the different components affecting medication adherence. Thus, the unit works on three research axes. Firstly, achieving a comprehensive understanding of patient adherence and self-management across several chronic disease models, and developing preventive approaches to nonadherence. Secondly, investigating the epidemiology of contradictory information on prescribed medications as perceived by chronic patients, and its impact on patient medication self-management. Lastly, analyzing how interprofessional collaborations could support the development of concerted information strategies in favor of a more efficient and secure drug use, particularly at the hospital-community interface.
Apply now
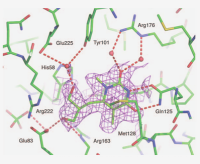
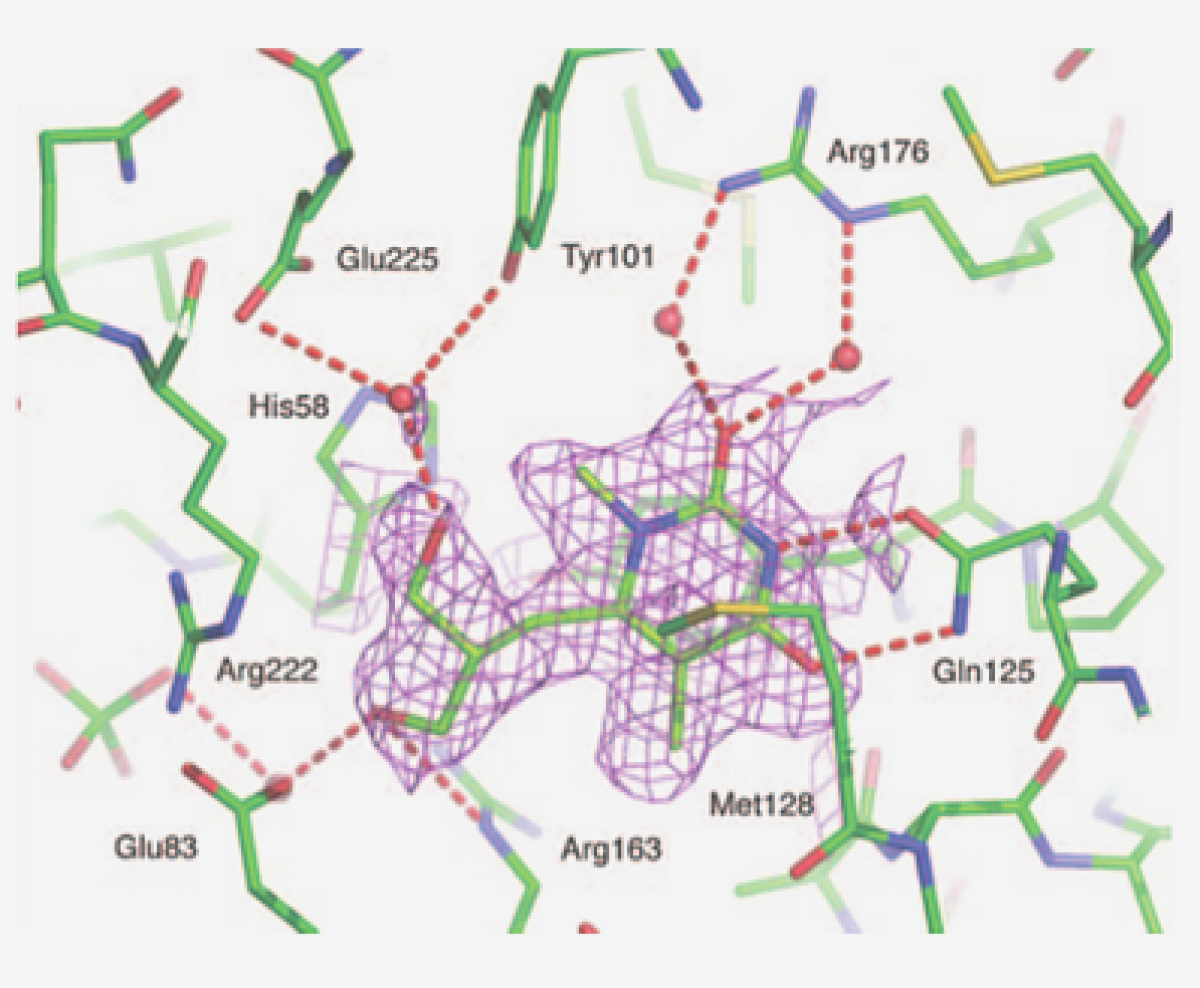
Pharmaceutical Biochemistry
The research of the Pharmaceutical Biochemistry group aims at addressing some of the challenges in taking a drug from the bench to the patient by integrating smart cutting edge science and technology using an interdisciplinary approach. Solving the twin problems of lack of efficacy and high toxicity goes through the selection of the right target, the right molecule and the appropriate delivery technology and formulation for the appropriate clinical trial. The research of the group tackles the issues related to choosing the right targets, and molecules. Research in the Pharmaceutical Biochemistry/Chemistry field led by Prof. Scapozza focuses on molecular recognition for a better understanding of ligand-macromolecule interactions to develop therapeutic strategies involving new chemical entities
Apply now
Biomedical and metabolomics analysis
As a member of the School of Pharmaceutical Sciences (University of Geneva, Switzerland), the group is developing new strategies for untargeted metabolomics analyses and specializes in the analysis of low molecular weight compounds in biological matrices. Since 2010, the group also focuses on developing chemometric approaches dedicated to the analysis of data produced by MS couplings, including capillary electrophoresis. Aspects of dimensionality reduction and multi-table analysis are addressed through collaborative projects in the fields of toxicology, biology, biochemistry, and pharmacology.
Apply now
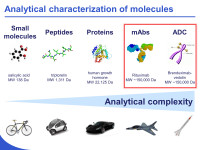
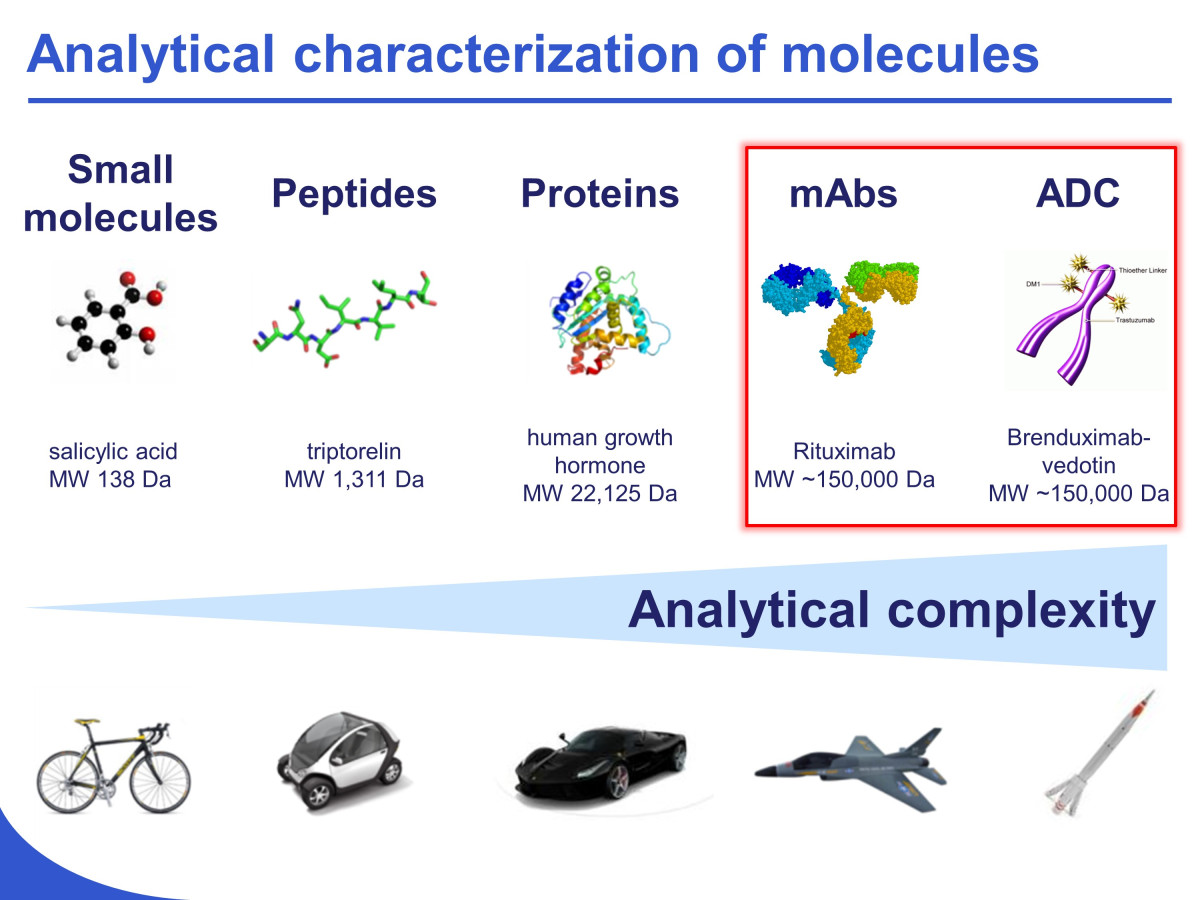
Development of innovative strategies for the analytical characterization of protein biopharmaceuticals
The characterization of therapeutic proteins, including monoclonal antibodies (mAbs) and antibody-drug conjugates (ADCs), is a tremendous challenge to state-of-the-art analytical technologies. Indeed, subtle changes in these large (> 150 kDa) molecules can have profound effects on efficacy and pharmacokinetic properties, thus it is important to have the ability to rapidly and accurately assess changes in the distribution of different isoforms (e.g., glycosylation, oxidation, deamidation, lysine truncation…) of such biomolecules. In our laboratory, we are developing some new analytical strategies based on liquid chromatography (LC) capillary electrophoresis (CE) and mass spectrometry (MS) for the characterization of therapeutic proteins.
Apply now

Hospital Pharmacy CHUV
Our hospital pharmacy group has very varied research activities: clinical pharmacy, drug formulation, pharmacoeconomics, pharmaceutical analysis, therapeutic education, development of parenteral nutrition especially in neonatology. The final goal of all our research is to optimize drug treatments and to warrant the safest and most efficient medication process and drug use in hospital, from industrial supply to patient administration.
Apply now
Pharmacogenomics and precision medicine
The research group's work focuses on investigating genetic determinants of variability in therapeutic response (pharmacogenomics). We study gene-environment-pathology interactions, especially at the pharmacokinetic level, including drug metabolism and transport. Phenoconversion, the discordance between predicted and actual phenotypes, is assessed using the Geneva cocktail. We evaluate genetic vulnerability to drug interactions and develop in vitro, in vivo, and in silico tools to measure and predict drug response. A key focus is optimizing PBPK modeling for personalized prescriptions and progressively integrating phenotypic and pharmacodynamic measures, with future use of machine learning algorithms.
Apply now

Improving natural products chemical biodiversity by enzymatic biotransformation
Natural products (NP) have been the starting point for the development of many therapeutic drugs throughout history. The classic way to obtain bioactive natural products is the fractionation of an active natural extract. Bioactive NP were also obtained by biological methods such as biotransformation reactions. We are building NP banks obtained by enzymatic biotransformation that could be used for future screening campaigns. We use a mixture of enzymes produced by a given microorganism to enrich the chemical diversity of the compounds generated. As substrates for biotransformation reactions, NPs with a wide variety of scaffolding are chosen. The compounds obtained are evaluated using different in vitro biological tests (antifungal, antibacterial, antiparasitic and cytotoxic).
Apply now
Laboratory of Metabolic Health
Our research bridges several disciplines: metabolism, immunology, bioinformatics, microbiology, physiology, tumor and systems biology. The interests include immunometabolism and roles of the adipose tissue and gut microbiota in regulation of metabolism and tumor growth. Using state-of-art technologies and mouse models, in vitro systems, cohorts of human patients, and bioinformatics, we aim at developing approaches and pharmaceutical formulations for treatment of metabolic diseases including diabetes, obesity and osteoporosis. Our work on the link between the gut microbiota and adipose tissue browning pioneered this research field, and paved a way for manipulating the microbial composition, and the modes of its signalling to the host for therapeutic purposes.
Apply now

Data Analytics
Data analytics can be described as a set of procedures including data processing, organization and analysis for decision making with controlled risk based on evidence provided by data. The aim of the Data Analytics Lab is to contribute to the development of new methodologies for data analysis and decision making that take into account the big data environment with massive amount of complex data. We focus on fundamental developments that make use of the latest advances in statistics and computer sciences, in particular machine learning. For better added-value and impact, we also collaborate, in an interdisciplinary spirit, with researchers in different fields.
Apply now

Science and practice of rational medication use in real life settings
Which drugs to which patient and at which dose? The research interests of the group of clinical pharmacy sciences are the improvement of knowledge on drugs and on the optimization of practice in real life setting. The research fields are based on (1) Pharmacometrics - the science of quantification of drugs’ pharmacokinetic and pharmacodynamic effects to aid efficient drug development and dosage individualization (2) Pharmacoepidemiology - the application of the science of epidemiology to the study of the safety, effectiveness and uses of drugs, focusing in particular on vulnerable populations (3) Clinical Pharmacy - the evaluation of specific approaches for drug use and the development of tools and guidelines for clinical practice.
Apply now

Drug combinations in personalized medicine
The overall aim of the Molecular Pharmacology Group’s research is the discovery of optimal drug combinations, ODCs, for the treatment of complex diseases, mainly cancer. We use a phenotypic statistics-based technology combined with data modeling to identify ODCs with a minimal in vitro experimental effort and to subsequently translate them to in vivo models. The major clinical relevance of this strategy are: (i) the approach is personalized, (ii) ODCs may be applicable to patients that failed conventional treatment, (iii) ODCs can be quickly adapted during the course of treatment addressing temporal tumor heterogeneity. Moreover, our strategy uses fundamental research to reveal the ODC action mechanisms. The latter may identify novel signaling pathways or reveal mechanisms of resistance.
Apply now

Theranostic developments for diagnostic and therapeutic radiopharmaceutical
The theranostic principle in nuclear medicine uses the same cancer target for both diagnostic and therapy, with a molecule radiolabelled differently or administered in different dosages. The imaging-based diagnostic offers valuable insights into anticipating the likelihood of tumour response to a specific treatment, enabling a patient-tailored radioligand therapy based on the individual molecular characteristics of the tumour. Clinically established theranostic strategies combine diagnostic using positron or gamma-emitters and therapy using alpha or beta-emitters for precise radiation delivery. Through a complete production and validation pipeline, our group aims at developing radioisotope-based pharmaceuticals as theranostic pairs for novel cancer molecular targets.
Apply now

Hospital Pharmacy HUG
The group is specialised in hospital pharmacy, and very varied research activities are developed to optimize drug treatments at hospital and promote a smooth continuity of care. The best efficacy/safety ratio of drug use is our main objective, as well as an efficient and safe process organization, with a full traceability of drugs from arrival to the hospital until administration to patients. To reach these goals, research activities covers different fields of hospital pharmacy practice: development of hospital pharmaceutical forms, pharmaceutical analysis, pharmacoeconomics, clinical use of drugs, information technologies, risk management and education through innovative pedagogic approaches (e-learning, simulation, …).
Apply now

Cellular therapy and transplantation
Our research group investigates immunological mechanisms driving the efficacy and toxicities of adoptive cell therapies, including allogeneic hematopoietic stem cell transplantation (HSCT) and chimeric antigen receptor (CAR) T cells. In HSCT, we study preclinical models and patient samples to understand relapse and graft-versus-host disease (GvHD), aiming to discover predictive biomarkers and develop preventive strategies using regulatory cells like Tregs and iNKT cells. For CAR T cells, we focus on mechanisms of resistance, combination therapies to enhance efficacy, and novel allogeneic CAR-based therapies for B-cell, T-cell, and myeloid malignancies.
Apply now
Pharmacognosy
The pharmacognosy research unit is focused on the search for cancer chemopreventive and antiparasitic natural products. In these areas, the development of new and better drugs remains a principal need. As established by ample precedent, nature provides broad chemical diversity. The in vitro activity of plant extracts and pure natural products is evaluated and then mechanistic studies are pursued with the most promising ones. The absorption and the metabolism of pure compounds and phytopreparations are also being evaluated in vitro and in vivo.
Apply now

BioAnalytical Mass Spectrometry
Our group is committed to advancing mass spectrometry to uncover the secrets of regulatory nucleic acids. We excel in 'native' mass spectrometry, revealing intricate nucleic acid structures and their interactions with ligands and proteins. Our vision involves developing new approaches to characterize new therapeutic classes and push the boundaries of RNA modification analysis. Projects encompass instrument and method development (mass spectrometry, including ion mobility spectrometry and laser photodissociation), data interpretation, molecular modeling, multiple biophysical approaches, and applications in biology and pharmacy. The PI commits to provide a research environment fostering creativity, cutting-edge research, a multidisciplinary and multicultural playground, and career support.
Apply now
Biopharmaceutical Sciences
Biopharmaceutical Sciences examine interactions of drugs with biological systems contributing to their absorption, distribution, metabolism, elimination and toxicity. Expertise in the formulation of drugs, preparation of drug carriers, and profound knowledge of biological systems involved is mandatory. The group focuses on the understanding on complex drugs of biological and non-biological origin (non-biological complex drugs, NBCDs) such as therapeutic proteins, nanomedicines and vaccines using physico-chemical characterization, developing in vitro models representing tissues involved in absorption, metabolism and immune response, and by in vivo studies. We strive to optimize the interaction between complex drugs and biological systems in terms of an enhanced efficacy and safety.
Apply now
Translational Brain Biomarkers
Le groupe de recherche s'articule autour de deux axes en neurobiologie a) recherche fondamentale et b) recherche translationnelle intégrant les compétences de chercheurs en biologie, biochimie et chimie, de bio-informaticiens et de cliniciens. L'axe de recherche fondamentale cible la découverte de nouveaux mécanismes dans la neurotoxicité de certains xénobiotiques sur des modèles in vitro de la barrière hématho-encéphalique. L'axe de recherche translationnelle a comme objectif l'identification, le développement et l'implémentation de marqueurs diagnostiques et prognostiques de pathologies cérébrales tels que les accidents vasculaires cérébraux et les traumatismes crâniens.
Apply now